Comparative Phytochemical Analysis and Bioactivities of Leaf and Stem Extracts of Nigerian Indigofera spicata
| Received 04 Oct, 2025 |
Accepted 22 Feb, 2026 |
Published 31 Mar, 2026 |
Background and Objective: The chemical composition of medicinal plants can vary across geographical regions, influencing their therapeutic potential. Indigofera spicata, a species widely used in traditional medicine, remains underexplored in Nigeria. This study aimed to comparatively evaluate the phytochemical composition and biological activities of the leaf and stem extracts of I. spicata collected in Nigeria, to identify potential pharmacologically active compounds. Materials and Methods: Leaf and stem samples were extracted using methanol, ethyl acetate, and n-hexane solvents. Phytochemical constituents were screened using standard qualitative assays. Antimicrobial activity was assessed through susceptibility tests, Minimum Inhibitory Concentration (MIC), and Minimum Bactericidal Concentration (MBC) analyses. Antioxidant potential was determined by the 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging assay, while cytotoxicity was evaluated using the Brine Shrimp Lethality Assay (BSLA). Statistical analyses were performed using appropriate comparative tests with significance set at p<0.05. Results: The leaf extract contained tannins, flavonoids, and cardiac glycosides, whereas the stem extract contained saponins, alkaloids, steroids, and cardiac glycosides. The stem extract exhibited stronger antimicrobial activity (MIC 0.39 mg/mL; MBC 3.125 mg/mL) against Escherichia coli. It also demonstrated superior antioxidant potential (IC50 = 245.77 μg/mL) compared to the leaf extract (LC50 = 1081.33 μg/mL). Cytotoxicity results indicated higher potency for the stem extract (LC50 = 290 μg/mL) relative to the leaf extract (LC50 = 507.5 μg/mL). Conclusion: The stem extract of Indigofera spicata collected in Nigeria exhibited superior antimicrobial, antioxidant, and cytotoxic properties compared to the leaf extract, suggesting it as a promising source of bioactive compounds for pharmaceutical and medicinal applications. Further isolation and characterization of active constituents are recommended to validate these findings.
| Copyright © 2026 Oloyede and Babatunde. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
The exploration of medicinal plants in traditional medicine has gained significant attention in recent years due to their potential as sources of bioactive compounds that can contribute to human health1,2. While traditional medicine has long utilized various plant species for their therapeutic properties, modern scientific research continues to validate these uses3. Among the myriads of plants studied, Indigofera spicata stands out due to its unique phytochemical profile and promising biological activities4.
Indigofera spicata is a ground-cover plant with butterfly-shaped flowers of various colors, ranging from red to orange to pink5. This plant species, also known as “creeping or trailing indigo”, belongs to the Fabaceae family. It is a perennial herbaceous plant commonly found in tropical and subtropical regions. Indigofera spicata is traditionally used for the treatment of meningitis, diarrhea, stomachache and diabetes6. It was also used for ground cover and erosion control in coffee, oil palm, rubber, sisal and tea plantations5. Later on, it was not used for the same purposes because of its CNS toxicity to animals, which could result in heir death4. Even though the plants of the genus Indigofera are widely recognized for yielding indigo dyes, previous research work demonstrated their antioxidant, antimicrobial, antispasmodic, hepatoprotective, and inhibitory properties7. Moreover, recent phytochemical analysis demonstrated the presence of high levels of flavonoids, alkaloids, and tannins in Indigofera spicata, which were earlier recognized for their antioxidant properties. However, the flavonoids of this plant are recognized for their capability to scavenge 'free radicals' or 'reactive oxygen species' that could damage cells by inducing oxidative stress, a crucial event contributing to the pathogenesis of various diseases such as cancer. Other members of the Indigofera genus are known for their antimicrobial, anti-inflammatory and analgesic, and hepatoprotective effects, suggesting similar potential in Indigofera spicata. Studies have demonstrated that extracts from Indigofera spicata exhibit significant cytotoxic effects against several cancer cell lines. For instance, research has shown that its extracts can induce apoptosis (programmed cell death) in HT-29 colon cancer cells8. This suggests that the bioactive compounds present in this plant may serve as potential chemotherapeutic agents or adjuvants in cancer treatment.
Although Indigofera spicata has shown promise in preliminary cytotoxicity studies, the stems of Indigofera spicata are among the lesser-explored botanical resources with significant medicinal potential8. Furthermore, since it is believed that the chemical composition of natural resources varies by location9, it is expected that extracts of Indigofera spicata collected in other regions, including Nigeria, may possess unique chemical constituents and properties suitable for alleviating the burden of these diseases. Therefore, comprehensive investigations of its chemical composition and mechanisms of action are still needed. Understanding how this plant exerts its effects at the molecular level will provide valuable insights into its potential applications in cancer prevention and treatment. Therefore, investigating the antioxidant capacity, cytotoxicity, and chemical compositions of Indigofera spicata is essential not only for understanding its therapeutic potential but also for developing natural products that can complement existing treatments and enhance potential applications in pharmacology and medicine.
MATERIALS AND METHODS
Study area: This study was carried out at the University of Ibadan, Ibadan, Nigeria, between the year 2024 to 2025. The fresh leaves and stems of Indigofera spicata (Fig. 1) were collected from Akefa farm, Ipara Remo, Ogun State, Nigeria. The phytochemical analysis and the biological activities were done at the F102 laboratory of the Department of Chemistry and the Pharmacognosy laboratory, respectively.
Materials: The collected were identified and authenticated at Forestry Research Institute of Nigeria (FRIN), Oyo State with voucher number 1142078. The leaves and stems were air-dried under shade, separated, and roughly ground before extraction.
Extraction of plant’s sample: The dried leaves and stems samples of Indigofera spicata were ground, and 239.96 and 281.06 g of its leaf and stem were obtained, respectively. These were carefully poured into well-labelled aspirator bottles and macerated with methanol for 72 hrs. The mixtures were decanted and concentrated into a paste via distillation and dried using a vacuum oven at a constant temperature of 40°C.

|
Phytochemical screening of Indigofera spicata leaf and stem extracts: Saponins were tested following a previous study by Savithramma et al.10. As such, an aliquot portion of the sample (about 1 mL) was dissolved in 20 mL of distilled water. The resulting solution was shaken vigorously for a few minutes. Frothing, which persisted on warming, was then taken as evidence of the presence of saponins.
The presence of tannins was screened for by stirring the plant fraction (0.5 g) with 10 mL of distilled water, warmed, and filtered. A few drops of 10% Ferric Chloride (FeCl3) were added to 2 mL of the filtrate. A blackish blue precipitate or green-blackish precipitate indicates the presence of gallic tannins or catechol tannins, respectively10.
Alkaloids were screened using both Mayer’s and Wagner’s tests. For the Mayer’s test, 1.36 g of mercuric chloride, HgCl2 was weighed. The 5 g of potassium iodide was also weighed. The two compounds were dissolved in 100 mL distilled water. A few drops of Mayer’s reagent were added to 1 mL of the plants’ samples. A yellowish or white precipitate indicates the presence of alkaloids. For the Wagner’s test, 2.5 g of iodine was weighed. 12.5 g of potassium iodide, KI was also weighed, then dissolved in 250 mL distilled water. A 1 mL of dilute hydrochloric acid was added to 2 mL of the plants’ samples. A few drops of Wagner reagent were added to the mixture. The formation of orange-brown precipitate indicates the presence of alkaloids11,12.
Flavonoids were identified by adding a few drops of dilute sodium hydroxide solution to a portion of the aqueous filtrate of test extracts (1 mL) to give an intense yellow coloration, followed by the addition of concentrated tetraoxosulphate (vi) acid. A colorless precipitate indicates the presence of flavonoids.
Steroids were assessed using Salkowski’s test. As such, 2 mL of chloroform was added to 1 mL of plant extract, followed by 1 mL of concentrated tetraoxosulphate (vi) acid by the side of the test tube. A yellow coloration with green fluorescence indicates the presence of steroids.

|
|
The cardiac glycosides was screened followingg the Salkowiski’s test. Thus, 1 mL of chloroform was added to 1 mL of plant extract, followed by 2 mL of concentrated tetraoxosulphate (vi) acid. The mixture was shaken, and the presence of reddish-brown precipitate indicates the presence of cardiac glycosides13.
Antimicrobial analysis: Antimicrobial activity was comprehensively assessed through susceptibility testing, determination of Minimum Inhibitory Concentration (MIC), and Minimum Bactericidal Concentration (MBC). For the susceptibility testing, sterile Mueller-Hinton Agar (20 mL) was aseptically dispensed into petri dishes, dried, and inoculated with a 10-5 dilution of an 18-hour broth adjusted to the McFarlan standard using the spread plate method. Wells (6 mm diameter) were bored into the inoculated agar, and different concentrations of Indigofera spicata plant extract were added (Fig. 2). Ciprofloxacin (5 μg) served as a positive control, and dimethyl sulfoxide (solvent) as a negative control. Plates were incubated at 37°C for 24 hrs after allowing 1 hr for extract diffusion, and diameters of zones of inhibition were measured in mm.
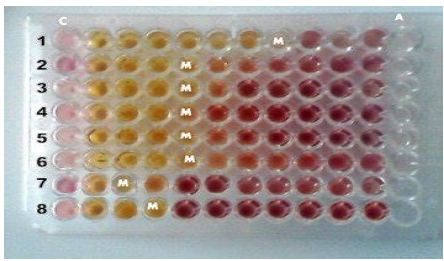
The Minimum Inhibitory Concentration (MIC) was determined using the broth microdilution method in 96-well plates (Fig. 3). Extract samples of Indigofera spicata were dissolved in double-strength Tryptone Soya Broth (MERK) to an initial concentration of 2.5 mg/mL, then serially diluted to obtain concentrations ranging from 1.25 mg/mL to 0.01953125 mg/mL. Ciprofloxacin (10 μg/mL stock) was similarly diluted for reference. After initial incubation and observation for growth, 10 μL of 0.2 mg/mL p-INT solution [piodonnitrotetrazolium violet] was added, and plates were further incubated at 37°C for 30 min; a color change from yellow to pinkish-red indicated bacterial/microbial growth. For the Minimum Bactericidal
Concentration (MBC), 10 μL of the microorganism was inoculated into each microplate well and incubated at 37°C for 24 hrs. The least concentrations that showed no growth or turbidity were then streaked onto Nutrient Agar (N.A.), and the lowest concentration with no trace of growth was considered the MBC.
Assessment of antioxidant activity: The antioxidant potential of Indigofera spicata extracts was evaluated by assessing their radical scavenging activity using 1,1-diphenyl-2-picrylhydrazyl (DPPH). The DPPH is a stable free radical characterized by its deep violet color, absorbing at approximately 517 nm in methanol solution. When DPPH interacts with a hydrogen atom-donating substrate, it is reduced, resulting in the loss of its violet color. To monitor this, 1 mL of Indigofera spicata sample extract was diluted with 1 mL of 0.3 mM DPPH solution. After 30 min, the absorbance was measured at 517 nm. The percentage inhibition of the DPPH radical was calculated using Eq. 1:
| (1) |
where, Abr is the absorbance control and Aar is the absorbance of the sample’s reaction.
Cytotoxicity investigations: The cytotoxicity investigation was carried out using the Brine Shrimp Lethality Assay (BSLA). Methanolic crude extracts of the leaves and stems of Indigofera spicata and the leaves of were subjected to the Artemia salina lethality assay. The larvae of brine shrimp were exposed to various concentrations of the extracts (31.25-1000 μg/mL) at room temperature for 24 hrs, after which the total number of living nauplii was counted and brine shrimp lethality (BSL) was determined using Eq. 2 14:
| (2) |
where, T is total number of larvae exposed to extracts and S is the total number of living nauplii. The experiment was done in triplicate.
RESULTS AND DISCUSSION
Yield of the crude extract: Table 1 presents the yield of the plant’s leaf and stem. The result obtained shows that the yield of the leaf extract exceeds the stem extract by a factor of 0.2.
Phytochemical analysis: Table 2 presents the result of the phytochemical analysis of the leaf and stem of Indigofera spicata obtained in this study. The phytochemical analysis was conducted for the methanol, n-hexane, and etylacette extractions. The present study revealed that the leaf extract contains tannins, flavonoids, and cardiac glycosides, while saponins, alkaloids, and steroids were not present. The stem extract contains saponins, alkaloids, steroids, and cardiac glycosides, while tannins and flavonoids were not found.
Antimicrobial activity: The antimicrobial activity of the leaf and stem extracts of the Indigofera spicata examined in this study has been investigated based on the susceptibility test, MIC, and MBC against several organisms. Table 3 presents the result obtained for the antimicrobial susceptibility test conducted on the leaf and stem extracts of Indigofera spicata. The results observed show that there were significant differences between the extract concentrations and the plants’ parts used on the susceptibility of the test microbes. For the three concentrations considered, the suscepeptibility zones against the test organisms range from 10 to 18 and 10 to 30 for the leaf and stem extracts of Indigofera spicata, respectively.
| Table 1: | Percentage yield of the crude extract | |||
| Plant sample | Weight of sample (g) | Weight of crude extract (g) | Yield of the extract (%) |
| Indigofera spicata leaf | 239.96 | 20.96 | 8.7 |
| Indigofera spicata stem | 281.06 | 23.76 | 8.5 |
| Table 2: | Phytochemical analysis of the leaf and stem extracts of Indigofera spicata | |||
| Leaf extract | Saponins | Tannins | Alkaloids | Flavonoids | Steroids | Cardiac glycosides |
| IS MeOH | - | + | - | + | - | + |
| IS n-Hexane | - | + | - | + | - | + |
| IS ETA | - | + | - | + | - | + |
| Stem extract | Saponins | Tannins | Alkaloids | Flavonoids | Steroids | Cardiac glycosides |
| IS MeOH | + | - | + | - | + | + |
| IS n-Hexane | + | - | + | - | + | + |
| IS ETA | + | - | + | - | + | + |
| +: Present -: Absent, IS: Indigofera spicata, MeOH: Methanol and ETA: Ethylacetate | ||||||
| Table 3: | Analysis of antimicrobial susceptibility test at various concentration (mg/mL) | |||
| IS Leaf | IS Stem | Control | Control | |||||
| Organism | 100 | 50 | 25 | 100 | 50 | 25 | Cipro | Ket |
| S.a | 0 | 18 | 0 | 14 | 12 | 10 | 18 | NA |
| B.s | 0 | 0 | 0 | 18 | 17 | 14 | 20 | NA |
| E.c | 0 | 0 | 0 | 30 | 26 | 21 | 24 | NA |
| Ps.a | 0 | 0 | 0 | 20 | 16 | 0 | 22 | NA |
| C.a | 12 | 10 | 0 | 14 | 0 | 0 | 14 | 18 |
| T.r | 14 | 10 | 0 | 0 | 0 | 0 | 0 | 12 |
| S.a: Staphylococcus aureus, B.s: Bacillus substilis, E.c: Escherichia coli, P.a: Pseudomonas aeruginosal, C.a: Candida albicans, T.r: Tricophyton rubrum, Cipro: Ciprofloxacin. Ket: Ketoconazole and IS: Indigofera spicata | ||||||||
| Table 4: | Minimum inhibitory concentration and minimum bactericidal concentration assays in mg/mL | |||
| IS Leaf | IS Stem | Control | |||||||
| Organism | MIC | MBC | MIC | MBC | B ONLY | B AND I | B AND E | Cipro (ug\mLs) | Ket (%) |
| S.a | >50 | >50 | 6.25 | 25 | - | - | + | 5 | NA |
| B.s | >50 | >50 | 12.5 | 12.5 | - | - | + | 10 | NA |
| E.c | >50 | >50 | 0.39 | 3.125 | - | - | + | >10 | NA |
| Ps.a | >50 | >50 | 25 | 25 | - | - | + | 10 | NA |
| C.a | >50 | >50 | 50 | 50 | - | - | + | NA | 0.25 |
| T.r | 50 | >50 | >50 | >50 | - | - | + | NA | 1 |
| Org: Organisms, B: Broth, I: Isolate, E: Extract, MIC: Minimum inhibitory concentration, MBC: Minimum bactericidal concentration S.a: Staphylococcus aureus, B.s: Bacillus substilis, E.c: Escherichia coli, P.a: Pseudomonas aeruginosal, C.a: Candida albicans and T.r: Tricophyton rubrum | |||||||||
| Table 5: | DPPH scavenging activivity at different concentrations of methanol crude extract | |||
| Conc (μg/mL) | IS Leaf (%) | IS Stem (%) | AA Std (%) |
| 500 | 61.70±0.13 | 61.70±0.13 | 96.40±0.02 |
| 250 | 27.58±0.07 | 47.35±0.09 | 79.95±0.08 |
| 125 | 16.90±0.04 | 40.21±0.17 | 55.30±0.04 |
| 62.5 | 13.47±0.25 | 33.65±0.18 | 36.12±0.02 |
| 31.25 | 6.26±0.06 | 27.71±0.18 | 24.42±0.03 |
| IC50 (μg/mL) | 1081.33±8.88e | 245.77±1.24d | 90.96±0.12c |
| AA Std: Ascorbic acid standard, IS: Indigofera spicata, Values represent Mean±SEM, values with different alphabets superscript are significantly different | |||
Table 4 presents the results of the minimum inhibitory and minimum bactericidal concentration tests conducted on the leaf and stem extracts of Indigofera spicata. Asides from Tricophyton rubrum (T.r), the leaf extracts had the highest maximum inhibitory and maximum bactericidal concentrations (>50) compared to the stem (MIC ranging from 0.39 to 50 and MBC ranging from 3.125 to 50).
| Table 6: | Average death of artemia salina at different concentrations of methanol crude extract | |||
| Average death of artemia salina concentration (μg/mL) | ||||||
| Plant sample | 1 | 10 | 100 | 500 | 1000 | LC50 (μg/mL) |
| IS Leaves | 0 | 1 | 1 | 2 | 5 | 507.5 |
| IS Stem | 1 | 2 | 3 | 3 | 7 | 290 |
| (-ve control) | 0 | 0 | 1 | 1 | 1 | - |
| (+ve control) | ||||||
| Potassium dichromate | 2 | 9 | 10 | 10 | 10 | 8.369 |
| IS: Indigofera spicata | ||||||
Antioxidant activity: Table 5 presents the results of the antioxidant activities of the leaf and stem extract of the Indigofera spicata obtained in this study. The result revealed that there were significant differences between the extract concentrations and the plants’ parts used for the DPPH scavenging activity. Similar to the previous studies15,16, it was generally observed that the DPPH scavenging activity increases as the concentration of the sample extract increases.
Cytotoxicity and activity: The result of the cytotoxicity assay of the leaf and stem extracts of Indigofera spicata obtained in this study is presented in Table 6. The leaf shows an average death of 0 and 5, while the stem shows an average death of 1 and 7 at a concentration of 1 and 1000 μg/mL, respectively. This implies a concentration-dependent increment in the mortality rate of the brine shrimp, as similarly indicated in other studies16,17.
The result of the phytochemical analysis obtained in this study (Table 2) was revealed as a function of the type and polarity used. This agreed with the previous studies18,19. It was observed that the leaf extracts contain tannins, flavonoids, and cardiac glycosides, while the stem extract contains saponins, alkaloids, steroids, and cardiac glycosides. Due to their multiple biological functions, these phytochemicals receive particular attention in drug discovery, thereby improving the field of medicinal research4. These include antihypertensive, antiarrhythmic, antimalarial, anticancer, antioxidant, neuroprotective, antitumor, antibacterial, antiviral, antiangiogenic, antimicrobial, analgesic, anti-inflammatory, and anti-proliferative properties20,21. Similar to this study, a previous study also revealed the presence of tannins, flavonoids, and cardiac glycosides in the leaf extrac6 and saponins, alkaloids, and steroids in the stem extract22.
Over the past few decades, efforts have accelerated to discover plant-based antimicrobials as bacteria develop resistance to antibiotics and new clinically effective antibiotics are not on the horizon23. Screening of raw materials from plants has been a subject of research to identify new natural compounds to replace synthetics. This is because plants produce numerous secondary metabolites possessing antimicrobial activities to protect themselves against microbial pathogens23,24. These metabolites have been useful in pharmaceuticals as antimicrobial agnets25. In this study, for all the concentrations, the leaf extracts show no antimicrobial activity against the B.s., E.c., and Ps.a. organisms, while T.r. the stem extracts show no antimicrobial activity against the T.r. organism (Table 3). The effectiveness of the stem over the leaf extract, for most of the organisms, is likely due to the presence of saponins, alkaloids, and steroids in the stem as compared to the leaf extract. Similar to the previous studies, the antimicrobial activity increased with an increase in concentration16,26.
The documented antibacterial mechanisms of medicinal plants on microbes were prevention of cell wall synthesis, accumulating in bacterial membranes causing energy exhaustion, or disrupting the cell membrane permeability, which had an effect of permeability increase and cellular constituents loss, disruption of membranes, and changing the structure and function of critical cellular constituents, causing mutation, cell damage, and death27. The inhibition and death are often quantified by MIC and MBC, which are, respectively, the least concentration of a substance for which microbial growth cannot be detected
by visual or spectrophotometric means in liquid culture and the lowest concentration of a substance capable of killing at least 99.9% of the initial inoculum of bacteria on agar28. The result of the MIC and MBC (Table 4) showed that the antibacterial activity of the stem was more effective when compared to the leaf extract26. The effectiveness was more pronounced against the Escherichia coli with a minimum inhibition and minimum bactericidal concentrations of 0.39 and 3.125, respectively. An antibacterial effect is said to be bactericidal when the ratio of the MBC to MIC is ≤4 and bacteriostatic if the ratio is >4. Considering the stem extract, which is the most effective against the tested organisms, the effectiveness is bactericidal, except for the Escherichia coli, for which the effectiveness is bacteriostatic (MBC/MIC 8.01). This tolerance in Escherichia coli is consistent with its highly regarded set of intrinsic and acquired mechanisms of resistance, which include the production of β-lactamase enzymes, the formation of protective capsules and biofilms, and the activity of efflux pumps that expel antimicrobial agents29. The ability of Escherichia coli to employ such sophisticated defenses against a wide range of chemotherapeutic agents had underscored its significant burden in human and veterinary medicine30. This is therefore not unconnected to the bacteriostatic, rather than bactericidal, activity of the Indigofera spicata stem extract observed in the current study.
An imbalance between the accumulation and production of free radicals (such as hydroxyl radicals, superoxide radicals, hydrogen peroxide, and singlet oxygen) in biological systems and the inability to detoxify them often results in oxidative stress, which can occur through several factors such as infection, inflammation, alcohol intake, smoking, and radiation exposure, among others, and can interfere with and damage nucleic acids, proteins, and lipids31,32. It has been investigated that oxidative stress is linked to several health conditions such as cancer, cardiovascular diseases, neurological diseases, respiratory diseases, rheumatoid arthritis, kidney diseases, and sexual maturation, among others33. Antioxidants have been reported to play an important role in counteracting oxidative stress through neutralization of free radicals33. In this study, the antioxidant activities of the leaf and stem extracts of Indigofera spicata have been investigated using the DPPH free radical scavenging assay. Several studies have also investigated the antioxidant activities of plants’ extracts using the DPPH assay16,34-37. It was noted that the stem extract was more effective than the leaf extract at every concentration except 500 μg/mL, where they showed the same level of antioxidant activity (Table 5). This is further justified by an important metric, IC50, showing a value of 245.77 and 1081.33 μg/mL for the stem and leaf extracts, respectively. This difference is not unconnected to the divergent phytochemical profiles between the plant parts34. As the search for natural antioxidants like herbs, spices, vegetables, and fruits in mitigating the adverse effects associated with the synthetic antioxidants remains a growing interest in biomedical research35, the natural antioxidant activity demonstrated in this study positions Indigofera spicata extracts, particularly the stem, as a promising potential for pharmaceutical applications.
Plant-derived phytochemicals have shown cytotoxic effects by inhibiting cell proliferation and inducing apoptosis against cancer cells38. The insight into the cytotoxic potential of the leaf and stem extracts of Indigofera spicata obtained in this study has been presented in Table 6. The cytotoxicity effects demonstrated can be attributed to the presence of phytochemicals presented in Table 2. The leaf extract showed relatively low toxicity, having an LC50 value of 507.5 μg/mL. At the highest concentration (1000 μg/mL), the mortality rate was still modest (5 deaths), suggesting that the leaf extract exhibits low cytotoxic activity. Conversely, the stem extract demonstrated a lower LC50 of 290 μg/mL, indicating higher toxicity. At the highest concentration, the stem extract caused the death of 7 out of 10 brine shrimp, suggesting that the stem extract contains compounds that may possess more potent cytotoxic properties compared to the leaf extract. According to Clarkson’s classification index, cytotoxicity of herbal extracts indicates that LC50 above 1000, 500 to 1000, 100 to 500, and below 100 μg/mL are considered as non-toxic, low toxic, moderately toxic, and highly toxic, respectively39. Based on this classification, the leaf extract shows low toxicity (LC50 value of 507.5 μg/mL), while the stem extract shows moderate toxicity (LC50 of 290 μg/mL). Since studies have shown that plant extracts with LC50 below 100 μg/mL, which are categorized as toxic, do not always indicate danger or outright toxicity toward humans17. It thus implies that the cytotoxicity effect of the leaf and stem extracts of the Indigofera spicata demonstratedin this study is safe for human uses. However, such a level of cytotoxicity, particularly for the stem, suggests Indigofera spicata extract potentially as an antitumor or anticancer agent, which can be explored in the pharmaceutical industries.
CONCLUSION
This study comparatively investigated the phytochemical content and antimicrobial, antioxidant, and cytotoxic activity of leaf and stem extracts of Indigofera spicata, collected in Nigeria. The results clearly show that the different fractions of the plant have diverse phytochemical content, which also influences their bioactivities. The stem extract, which uniquely contained saponins, alkaloids, and steroids, possessed much higher antimicrobial, antioxidant, and cytotoxic activity than the leaf extract. Specifically, the stem extract exhibited strong inhibitory activity against the tested microorganisms, higher free radical scavenging activity, and higher toxicity against the brine shrimp lethality test. The enhancement in the efficacy of the stem extract makes it a superior source for the isolation of the therapeutic potential compounds, particularly for antimicrobial and antioxidant activities. The cytotoxicity observed in the stem extract (IC50 290 μg/mL) potentially reflects good antitumour activity. This study contributes to the scientific evidence of Indigofera spicata in medicine and identifies its stem as a possible source for future pharmacological studies and drug development. Future studies should focus on the separation and identification of the chemicals responsible for these activities and their molecular mechanisms of action.
SIGNIFICANCE STATEMENT
For several years, the demand for medications derived from natural products to mitigate the burdens of several diseases have extensively increased. This study investigated the leaf and stem extracts of Nigerian Indigofera spicata for their phytochemical properties and antibacterial, antioxidant, and cytotoxicity activities. The phytochemical screening of the leaf extract revealed the presence of tannins, flavonoids, and cardiac glycosides, while the stem extract contained saponins, alkaloids, steroids, and cardiac glycosides. The presence of these phytochemicals provides antibacterial, antioxidant, and cytotoxicity activities, which were mostly pronounced in the stem extract relative to the leaf extract. This study, therefore, projects Indigofera spicata extracts as an additional source of natural products that could serve as antibacterial, antioxidant, and cytotoxicity agents in pharmaceutical industries for the production of therapeutic medications to mitigate the existing burdens of cancer, cardiovascular diseases, neurological diseases, respiratory diseases, rheumatoid arthritis, kidney diseases, and sexual maturation, among others.
ACKNOWLEDGMENTS
The authors would like to acknowledge the effort of Dr. Abayomi M. Olaosun of the Department of Physics and Science Laboratory Technology of Abiola Ajimobi Technical University, Ibadan, Nigeria, and Mr. Olubunmi Mada of the Department of Pharmacognosy, University of Ibadan, Nigeria, for their contributions towards the success of this research work.
REFERENCES
- Coopoosamy, R., K. Singh, K. Naidoo and D.S. Nadasan, 2023. The role of phytomedicine: Bridging the gap between the past, present, and future. J. Med. Plants Econ. Dev., 7.
- Dar, R.A., M. Shahnawaz, M.A. Ahanger and Irfan ul Majid, 2023. Exploring the diverse bioactive compounds from medicinal plants: A review. J. Phytopharmacology, 12: 189-195.
- Salmerón-Manzano, E., J.A. Garrido-Cardenas and F. Manzano-Agugliaro, 2020. Worldwide research trends on medicinal plants. Int. J. Environ. Res. Public Health, 17.
- Mouafon, I.L., G.L.M. Tiani, B.Y.G. Mountessou, M. Lateef and M.S. Ali et al., 2021. Chemical constituents of the medicinal plant Indigofera spicata Forsk (Fabaceae) and their chemophenetic significance. Biochem. Syst. Ecol., 95.
- Pérez, L.B., J. Li, D.D. Lantvit, L. Pan and T.N. Ninh et al., 2013. Bioactive constituents of Indigofera spicata. J. Nat. Prod., 76: 1498-1504.
- Birru, E.M., M. Abdelwuhab and Z. Shewamene, 2015. Effect of hydroalcoholic leaves extract of Indigofera spicata Forssk. on blood glucose level of normal, glucose loaded and diabetic rodents. BMC Complementary Altern. Med., 15.
- Taj Ur Rahman, M.A. Zeb, W. Liaqat, M. Sajid, S. Hussain and M.I. Choudhary, 2018. Phytochemistry and pharmacology of genus Indigofera: A review. Rec. Nat. Prod., 12: 1-13.
- Pérez, L.B., J. Li, H. Chai, L. Pan and T.N. Ninh et al., 2012. Potential cancer chemotherapeutic agents from Indigofera spicata. Planta Med., 78.
- Akkawi, M., S. Jaber, S. Abu-Lafi, M. Qutob, Q. Abu-Rmeleh and P. Lutge, 2014. HPLC separation and in vitro antimalarial studies of Artemisia annua plants from two different origins: Cameroon versus Luxembourg. MalariaWorld J., 5.
- Savithramma, N., M.L. Rao and D. Suhrulatha, 2011. Screening of medicinal plants for secondary metabolites. Middle-East J. Sci. Res., 8: 579-584.
- Puri, B. and A. Hall, 1998. Phytochemical Dictionary: A Handbook of Bioactive Compounds from Plants. 2nd Edn., CRC Press, Boca Raton, Florida, ISBN: 9780203483756, Pages: 976.
- Evans, W.C. and D. Evans, 2009. Trease and Evans' Pharmacognosy. 16th Edn., Elsevier Health Sciences, Maryland, United States., ISBN: 978-0-7020-2933-2, Pages: 603.
- Silva, G.L., I.S. Lee and D.A. Kinghorn, 1998. Special Problems with the Extraction of Plants. In: Natural Products Isolation, Methods in Biotechnology, Cannell, R.J.P. (Ed.), Humana Press Inc., New Jersey, ISBN: 978-1-59259-256-2, pp: 343-363.
- Ferrante, C., L. Recinella, M. Ronci, G. Orlando and S. di Simone et al., 2019. Protective effects induced by alcoholic Phlomis fruticosa and Phlomis herba-venti extracts in isolated rat colon: Focus on antioxidant, anti-inflammatory, and antimicrobial activities in vitro. Phytother. Res., 33: 2387-2400.
- Okwute, S.K. and I.O. Ochi, 2023. Antimicrobial, antioxidant, anti-inflammatory and acute toxicity screening of leaf extracts of Morinda lucida (Rubiaceae). J. Biomed. Res. Environ. Sci., 4: 372-382.
- Alfuraydi, A.A., I.M. Aziz and F.N. Almajhdi, 2024. Assessment of antioxidant, anticancer, and antibacterial activities of the rhizome of ginger (Zingiber officinale). J. King Saud Univ. Sci., 36.
- Umaru, I.J., F.A. Badruddin, Z.B. Assim and H.A. Umaru, 2018. Cytotoxicity (brine shrimp lethality bioassay) of barringtoniaracemosa leaves, stem-bark and root extract. J. Biotechnol. Bioeng., 2: 45-50.
- Ibrahim, M., A.S. Idoko, A.I. Ganiyu, N. Lawal and P. Abu et al., 2023. Phytochemical analysis of hexane, chloroform, ethyl acetate, ethanol and aqueous extracts of Azanza garckeana leaf. Sahel J. Life Sci. FUDMA, 1: 25-31.
- Shofiqul Islam, M., S. Sana, M. Ehsanul Haque, S.M. Mushiur Rahman and Abdus Samad et al., 2021. Methanol, ethyl acetate and n-hexane extracts of Tragia involucrata L. leaves exhibit anxiolytic, sedative and analgesic activity in Swiss albino mice. Heliyon, 7.
- Olofinsan, K., H. Abrahamse and B.P. George, 2023. Therapeutic role of alkaloids and alkaloid derivatives in cancer management. Molecules, 28.
- Uzor, P.F., 2020. Alkaloids from plants with antimalarial activity: A review of recent studies. Evidence-Based Complementary Altern. Med., 2020.
- Musyimi, D.M., F.M. Kiema and G. Muinde, 2022. Phytochemical compounds and antimicrobial activity of extracts of Indigofera spicata (creeping indigo) against Candida albicans, Streptococcus mutans and E. coli. Bacterial Empire, 5.
- Bubonja-Šonje, M., S. Knežević and M. Abram, 2020. Challenges to antimicrobial susceptibility testing of plant-derived polyphenolic compounds. Arch. Ind. Hyg. Toxicol., 71: 300-311.
- Angelini, P., 2024. Plant-derived antimicrobials and their crucial role in combating antimicrobial resistance. Antibiotics, 13.
- Ogbuagu, O.O., A.O. Mbata, O.D. Balogun, O. Oladapo, O.O. Ojo and M. Muonde, 2025. Traditional medicinal plants as a source of new antimicrobial agents: Opportunities for scalable drug development. Int. Res. J. Modernization Eng. Technol. Sci., 7: 1476-1488.
- Belitibo, D.B., A. Meressa, T. Negassa, A. Abebe and S. Degu et al., 2025. In vitro antimicrobial and cytotoxic evaluation of leaf, root, and stem extracts of Solanum dasyphyllum and root and stem extracts of Dovyalis abyssinica. Front. Pharmacol., 16.
- Kang, C.G., D.S. Hah, C.H. Kim, Y.H. Kim, E.K. Kim and J.S. Kim, 2011. Evaluation of antimicrobial activity of the methanol extracts from 8 traditional medicinal plants. Toxicol. Res., 27: 31-36.
- Makade, C.S., P.R. Shenoi, B.A. Bhongade, S.A. Shingane, P.C. Ambulkar and A.M. Shewale, 2024. Estimation of MBC: MIC ratio of herbal extracts against common endodontic pathogens. J. Pharm. Bioallied Sci., 16: S1414-S1416.
- Nasrollahian, S., J.P. Graham and M. Halaji, 2024. A review of the mechanisms that confer antibiotic resistance in pathotypes of E. coli. Front. Cell. Infect. Microbiol., 14.
- Poirel, L., J.Y. Madec, A. Lupo, A.K. Schink, N. Kieffer, P. Nordmann and S. Schwarz, 2018. Antimicrobial resistance in Escherichia coli. Microbiol. Spectrum, 6.
- Azzi, A., 2022. Oxidative stress: What is it? Can it be measured? Where is it located? Can it be good or bad? Can it be prevented? Can it be cured? Antioxidants, 11.
- Pizzino, G., N. Irrera, M. Cucinotta, G. Pallio and F. Mannino et al., 2017. Oxidative stress: Harms and benefits for human health. Oxid. Med. Cell. Longevity, 2017.
- Aini, N. and A. Tilaqza, 2025. Evaluation of antioxidant activity of methanol extract of neem leaves (Azadirachta indica) using DPPH, ABTS, and FRAP methods. Sci. J., 4: 355-363.
- Farag, R.S., M.S. Abdel-Latif, H.H. Abd El Baky and L.S. Tawfeek, 2020. Phytochemical screening and antioxidant activity of some medicinal plants crude juices. Biotechnol. Rep., 28.
- Geleta, W.D., K.B. Gebru, A.A. Dessie, Y.A. Yusuf, G.H. Gebrewbet and B.T. WoldeMichae, 2025. Optimization of antioxidant extraction from Moringa oleifera seeds using response surface methodology: Phytochemical analysis and DPPH assay. J. Food Process. Preserv., 2025.
- Ilie, E.I., L. Popescu, E.A. Luță, A. Biță and A.R. Corbu et al., 2024. Phytochemical characterization and antioxidant activity evaluation for some plant extracts in conjunction with pharmacological mechanism prediction: Insights into potential therapeutic applications in dyslipidemia and obesity. Biomedicines, 12. h.
- Kudamba, A., G.S. Bbosa, A. Lugaajju, H. Wabinga and N. Niyonzima et al., 2025. Exploring the anticancer potential of R. tridentata extracts: A cytotoxicity study against human prostate cancer cell lines (LNCaP and DU145). Med. Oncol., 42.
- Isah, M., G. Zengin, W.N.A. wan Abdul Wahab, H. Abdullah and M.D. Sul’ain et al., 2024. Antioxidant, enzyme inhibition, toxicity, and molecular docking analysis of Melaleuca cajuputi leaf extract and fractions. Nat. Resour. Hum. Health, 4: 89-97.
- del Socorro, M.M.L, C.P. Bendoy and C.M.L. Dacayana, 2014. Cytotoxic effects of betel vine, Piper betle Linn. leaf extracts using Artemia salina Leach (brine shrimp lethality assay). J. Multidiscip. Stud., 3: 100-111.
How to Cite this paper?
APA-7 Style
Oloyede,
G.K., Babatunde,
T.H. (2026). Comparative Phytochemical Analysis and Bioactivities of Leaf and Stem Extracts of Nigerian Indigofera spicata. Trends in Pharmacology and Toxicology, 2(1), 36-46. https://doi.org/10.21124/tpt.2026.36.46
ACS Style
Oloyede,
G.K.; Babatunde,
T.H. Comparative Phytochemical Analysis and Bioactivities of Leaf and Stem Extracts of Nigerian Indigofera spicata. Trends Pharm. Toxicol. 2026, 2, 36-46. https://doi.org/10.21124/tpt.2026.36.46
AMA Style
Oloyede
GK, Babatunde
TH. Comparative Phytochemical Analysis and Bioactivities of Leaf and Stem Extracts of Nigerian Indigofera spicata. Trends in Pharmacology and Toxicology. 2026; 2(1): 36-46. https://doi.org/10.21124/tpt.2026.36.46
Chicago/Turabian Style
Oloyede, Ganiyat, K., and Tolulope H. Babatunde.
2026. "Comparative Phytochemical Analysis and Bioactivities of Leaf and Stem Extracts of Nigerian Indigofera spicata" Trends in Pharmacology and Toxicology 2, no. 1: 36-46. https://doi.org/10.21124/tpt.2026.36.46

This work is licensed under a Creative Commons Attribution 4.0 International License.



