Acute Toxicity of Bispyribac Sodium on the Survival of African Catfish (Clarias gariepinus) Juveniles and its Implications for Food Safety
| Received 24 Nov, 2025 |
Accepted 15 Jan, 2026 |
Published 31 Mar, 2026 |
Background and Objective: The increasing use of Bispyribac sodium in agriculture raises concerns about its potential toxicity to non-target aquatic species. However, information on its acute effects on Clarias gariepinus juveniles, a widely cultured food fish, remains limited. This study assessed the acute toxicity of Bispyribac sodium to C. gariepinus juveniles and examined associated changes in water physicochemical parameters. Materials and Methods: Juvenile C. gariepinus were exposed for 96 hrs to five concentrations of Bispyribac sodium (0.59, 0.88, 1.31, 1.97, and 2.95 mg/L) and a control (0.00 mg/L). Each treatment contained ten fish in replicated 70-L bowls filled with 20 L of water. Mortality was recorded upon cessation of gill movement and lack of response to gentle prodding. Water quality parameters (pH, dissolved oxygen, temperature, electrical conductivity, and total dissolved solids) were measured using a HANNA multiparameter meter. Mortality data were analyzed to estimate the 96 hrs LC50 using probit analysis, with treatment effects assessed by one-way ANOVA followed by Dunnett’s test (α = 0.05), and all analyses were conducted in SPSS v25. Results: Mortality increased with concentration, ranging from 20% at 0.59 mg/L to 75% at 2.95 mg/L, with no mortality in the control. The 96 hrs LC50 was 1.658 mg/L (95% CI: 1.204-2.289 mg/L), indicating a strong dose-dependent relationship (Probit = 2.1874×LogC+4.5189, R2 = 0.9552). Significant changes (p<0.05) occurred in pH (6.15-8.05), TDS (327.0-358.0 mg/L), EC (654.0-716.0 μS/cm), and DO (5.60-2.75 mg/L), while temperature remained stable. Fish at higher concentrations exhibited restlessness, weakness, and edge-settling behavior. Conclusion: Bispyribac sodium demonstrated considerable acute toxicity to C. gariepinus juveniles and induced notable alterations in water quality parameters. The species’ sensitivity suggests its suitability as a bioindicator of herbicide contamination. Careful regulation of Bispyribac sodium is recommended to minimize risks to aquatic health and food safety. Further studies are needed to refine safety thresholds for aquaculture environments.
| Copyright © 2026 Kwaghvihi et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Water, constituting approximately 70% of Earth’s surface, is essential for the survival and growth of all living organisms1. However, escalating human activities, including population growth, industrialization, agricultural fertilizer use, and anthropogenic pollution, have severely compromised water quality worldwide2-4. Natural water bodies accumulate impurities from diverse sources, such as rock weathering, soil leaching, atmospheric aerosol dissolution, mining, and runoff from metal-based fertilizers, leading to elevated concentrations of contaminants in freshwater reservoirs5. The Nigerian National Agency for Food and Drug Administration and Control (NAFDAC, 2011) estimates that one-third of global water sources are unfit for human consumption, posing significant risks of waterborne diseases and ecosystem disruption. Additionally, herbicide residues in water bodies can contaminate aquatic organisms like fish, which are critical food sources, raising concerns about food safety and human health risks6.
Water quality is governed by its physical, chemical, and biological properties, which exhibit wide variability and directly influence hydrobiological relationships and ecosystem metabolism1. Routine monitoring of these parameters is critical for ensuring potable water safety and preventing adverse health outcomes2. Among aquatic pollutants, herbicides represent a major threat due to their widespread agricultural application, which enhances crop yields but causes indiscriminate runoff into water bodies, endangering non-target organisms, including fish populations and potentially compromising the safety of fish consumed by humans7.
Toxicity studies evaluate the adverse effects of chemicals on biological systems, focusing on their chemical nature, interactions, and safety assessments. Toxicants are substances that induce harmful effects, which vary based on factors such as physical form (solid, liquid, gas), exposure duration (acute or chronic), administration route (dermal, oral, inhalation), and organism-specific variables like genetics and health status. Acute toxicity, defined as adverse effects from short-term exposure (typically ≤96 hrs), is commonly quantified by the median Lethal Concentration (LC50), representing the concentration required to cause 50% mortality in a test population8.
Bispyribac sodium, a sulfonylurea herbicide, is extensively used to control broadleaf weeds and sedges in rice paddies. While effective agronomically, its persistence in aquatic environments raises concerns about bioaccumulation and toxicity to non-target aquatic species, particularly economically important fish like the African catfish (Clarias gariepinus). This is particularly critical as Clarias gariepinus is a staple food fish in many regions, and herbicide residues may pose risks to human consumers through the food chain7. Juveniles of C. gariepinus are especially vulnerable due to their high metabolic rates and limited detoxification capacities, making them ideal bioindicators for ecotoxicological assessments. Despite its commercial significance, data on the acute toxicity of bispyribac sodium to C. gariepinus juveniles remain limited. This study addresses this gap by determining the 96 hrs LC50 values, providing essential baseline data for risk assessment, regulatory guidelines, and sustainable herbicide management in aquaculture-adjacent ecosystems while highlighting implications for food safety in fish consumption.
MATERIALS AND METHODS
Experimental site: The experiment was conducted at the Department of Fisheries and Aquaculture, Joseph Sarwuan Tarka University, Makurdi, Benue State, Nigeria, for the period of three months (March -May, 2024). The study was performed in a controlled laboratory environment to ensure consistent conditions throughout the experimental period.
Experimental fish: The research involved live African catfish juveniles (Clarias gariepinus) and was conducted in compliance with animal welfare and fish handling standards of the World Organization for Animal Health and the National Research Council. Ethical approval was obtained from the Department of Fisheries and Aquaculture of Joseph Sarwuan Tarka University, Makurdi, prior to commencement of the study. All protocols were designed to minimize stress and prevent harm.
One hundred and twenty Juvenile African catfish (Clarias gariepinus) were sourced from Oracle Fish Farm, Makurdi, Benue State, Nigeria. The fish were transported to the laboratory in oxygenated polyethylene bags. Before the experiment, the juveniles were acclimatized for seven days in 100 L fiberglass tanks filled with dechlorinated tap water. During acclimatization, fish were fed a commercial pelleted feed (Coppens, 2 mm, 40% crude protein) at 5% of their body weight twice daily (08:00 and 16:00). Feeding was discontinued 24 hrs before the start of the experiment to minimize metabolic waste interference
Experimental design and setup: A static acute toxicity bioassay was conducted to determine the 96 hrs median Lethal Concentration (LC50) of bispyribac-sodium (CAS Number: 125401-92-5, commercial formulation) on Clarias gariepinus juveniles. Bispyribac-sodium was procured from Oxnet Agrochemical Shop, High Level, Makurdi, Nigeria.
The experimental design consisted of a control and five treatment concentrations, each replicated three times, following a completely randomized design. Ten juvenile C. gariepinus (selected for uniform size and health) were randomly assigned to each replicate. The experiment was conducted in 70 L plastic bowls filled with 20 L of dechlorinated tap water. Based on preliminary range-finding tests, five concentrations of bispyribac-sodium were prepared: 0.59, 0.88, 1.31, 1.97, and 2.95 μL/L, with a control group (0.00 μL/L). The test solutions were prepared by diluting a stock solution of bispyribac-sodium in dechlorinated water, ensuring thorough mixing.
The experiment ran for 96 hrs under static conditions (no water renewal). Mortality was recorded at 24, 48, 72, and 96 hrs. A fish was considered dead when it exhibited no opercular movement and failed to respond to gentle prodding with a glass rod. Dead fish were promptly removed to prevent deterioration of water quality. The LC50 was calculated using probit analysis9.
Physicochemical parameters of test media: Water quality parameters were monitored daily to assess their influence on the test organisms and ensure consistency across treatments. The following parameters were measured:
| • | Dissolved oxygen (DO): Measured using a digital DO meter (model YSI Pro20), accuracy ±0.1 mg/L | |
| • | Temperature: Recorded using a Hanna multi-parameter water quality meter (model HI98194) | |
| • | pH: Determined using the same Hanna multi-parameter meter with a pH electrode (accuracy ±0.01) | |
| • | Total dissolved solids (TDS): Measured in mg/L using the Hanna multi-parameter meter | |
| • | Electrical conductivity (EC): Recorded in μS/cm using the Hanna multi-parameter meter |
Measurements were taken by immersing the calibrated electrode probes into the test media, with readings recorded after stabilization. All instruments were calibrated according to the manufacturer’s instructions before use. Water quality parameters were maintained within the optimal range for C. gariepinus (DO: 5.0-8.0 mg/L; temperature: 25-28°C; pH: 6.5-8.5) to minimize stress unrelated to the toxicant.
Statistical analysis: Mortality data were analyzed to determine the 96 hrs LC50 using probit analysis. Differences in mortality rates across concentrations were evaluated using One-way Analysis of Variance (ANOVA), followed by Dunnett’s post hoc test to compare treatment groups against the control (α = 0.05). Water quality parameters were analyzed for significant variations across treatments using ANOVA. All statistical analyses were performed using SPSS version 25.
RESULTS
Mortality and probit analysis of Clarias gariepinus juveniles exposed to bispyribac sodium herbicide: Juvenile Clarias gariepinus exposed to lethal concentrations of Bispyribac sodium herbicide exhibited symptoms of toxicosis, including uncoordinated movement, vertical swimming, gasping for oxygen, restlessness, loss of equilibrium, skin hemorrhage, and death. Mucus accumulation was observed on the body surfaces and gill filaments of deceased fish. As the concentration of Bispyribac sodium increased, fish displayed heightened restlessness, weakness, and a tendency to settle at the edges or bottom of the experimental bowls, indicating a dose-dependent response. Higher concentrations correlated with increased mortality rates.
|
|
| Table 1: | Mortality of Clarias gariepinus juveniles exposed to acute concentrations of bispyribac sodium | |||
| Treatment (mg/L) | No. of fish | Total mortality | % Mortality | Log concentration | Probit |
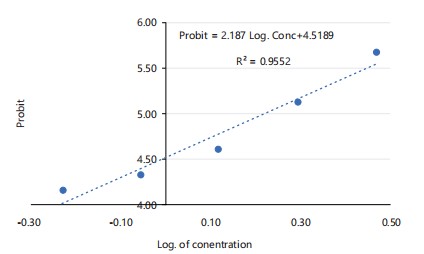
| 0 | 20 | 0 | 0 | 0 | 0 |
| 0.59 | 20 | 4 | 20 | -0.23 | 4.16 |
| 0.88 | 20 | 5 | 25 | -0.06 | 4.33 |
| 1.31 | 20 | 7 | 35 | 0.12 | 4.61 |
| 1.97 | 20 | 11 | 55 | 0.29 | 5.13 |
| 2.95 | 20 | 15 | 75 | 0.47 | 5.67 |
The highest mortality (75%) was observed in Treatment 6 (2.95 mg/L), while the lowest mortality (20%) was recorded in Treatment 2 (0.59 mg/L). No mortality was observed in the control treatment (0.00 mg/L) throughout the 96 hrs exposure period. The probit values, calculated based on the percentage mortality, indicate a clear relationship between herbicide concentration and mortality, supporting the use of probit analysis to estimate the lethal concentration (LC50) (Table 1).
From probit analysis (Fig. 1), the 96 hrs LC50 of Bispyribac sodium was calculated as 1.658 mg/L, with upper and lower confidence limits of 2.289 and 1.204 mg/L, respectively. The regression equation for the relationship between probit and log concentration was determined as Probit = 2.1874×Log(Concentration)+4.5189, with an R2 value of 0.9552. The high R2 value indicates a strong linear relationship, confirming that the mortality rate of Clarias gariepinus juveniles increased with increasing concentrations of Bispyribac sodium.
|
|
| Table 1: | Physicochemical parameters of experimental water during the 96 hrs acute toxicity test of Clarias gariepinus juveniles exposed to bispyribac sodium | |||
| Treatment (mg/L) | pH | Temperature (°C) | TDS (mg/L) | EC (μS/cm) | DO (mg/L) |
| 0 | 6.15±0.05a | 27.50±0.10 | 327.0±1.00a | 654.0±2.00a | 5.60±0.00a |
| 0.59 | 6.80±0.10b | 27.50±0.00 | 331.5±0.50b | 663.0±1.00b | 4.65±0.50b |
| 0.88 | 7.10±0.10c | 27.20±0.20 | 340.0±0.50c | 681.0±1.00c | 4.30±0.10c |
| 1.31 | 7.15±0.05c | 27.50±0.10 | 353.0±1.00d | 706.0±2.00d | 4.10±0.10c |
| 1.97 | 7.80±0.10d | 27.45±0.10 | 356.0±0.50e | 713.0±1.00e | 3.35±0.15d |
| 2.95 | 8.05±0.05d | 27.40±0.10 | 358.0±1.00e | 716.0±2.00e | 2.75±0.05e |
| p-value | 0 | 0.432 | 0 | 0 | 0 |
| Means in the same column with different superscripts (a,b,c,d,e) differ significantly (p 0.05) | |||||
|
|
Physicochemical parameters of the experimental water during the acute toxicity test: The physicochemical parameters of the test solutions during the 96 hrs acute toxicity test of Clarias gariepinus juveniles exposed to Bispyribac sodium are presented in Table 2. Parameters measured included pH, temperature (°C), total dissolved solids (TDS, mg/L), electrical conductivity (EC, μS/cm), and dissolved oxygen (DO, mg/L). Statistical analysis (ANOVA, p<0.05) revealed significant differences in pH, TDS, EC, and DO between the control (0.00 mg/L) and treatments, while temperature showed no significant variation (p = 0.432).
|
|
The control had the lowest pH (6.15±0.05), which increased with higher Bispyribac sodium concentrations, reaching 8.05±0.05 in Treatment 5 (2.95 mg/L). Similarly, TDS and EC were lowest in the control (327.0±1.00 mg/L and 654.0±2.00 μS/cm, respectively) and highest in Treatment 5 (358.0±1.00 mg/L and 716.0±2.00 μS/cm, respectively). Conversely, DO decreased with increasing herbicide concentration, from 5.60±0.00 mg/L in the control to 2.75±0.05 mg/L in Treatment 5. Temperature remained stable across all treatments, ranging from 27.20±0.20°C to 27.50±0.10°C.
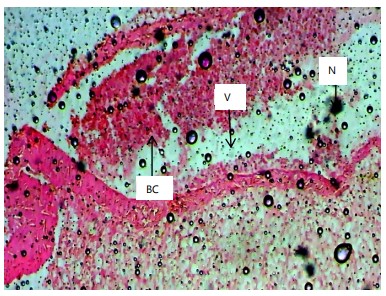
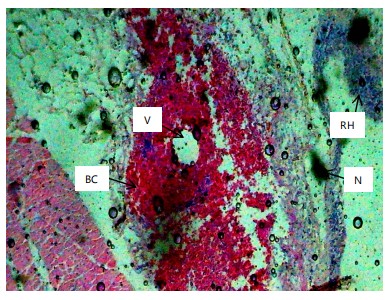
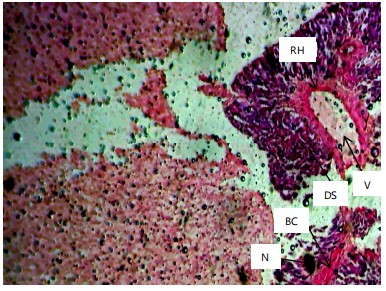
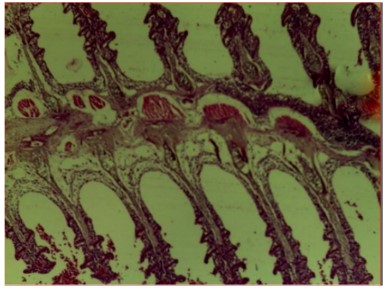
Histopathological examination of the liver and gill tissues of Clarias gariepinus juveniles revealed significant and concentration-dependent alterations following exposure to Bispyribac sodium herbicide, compared to the control groups, which displayed normal tissue architecture.
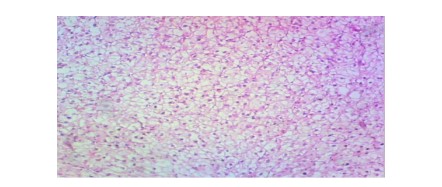
In the liver (Fig. 2-7), the control group (0.00 mg/L) exhibited normal hepatic structure with intact hepatocytes. Exposure to Bispyribac sodium induced a range of lesions, beginning at the lowest concentration of 0.59 mg/L with blood congestion, vacuolation, and necrosis. The severity and prevalence of these pathologies increased with concentration. At 1.97 mg/L, rupture of hepatocytes (RH) was observed, and the highest concentration of 2.95 mg/L resulted in the most severe damage, characterized by the addition of sinusoid dilation (DS) alongside the other noted lesions.
|
|
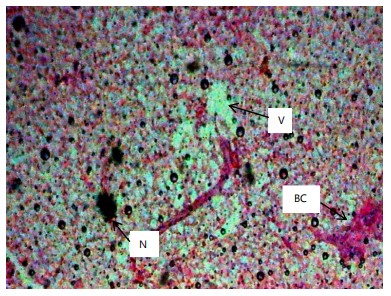
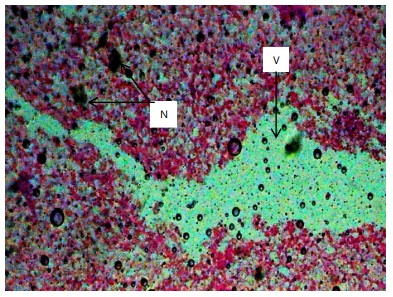
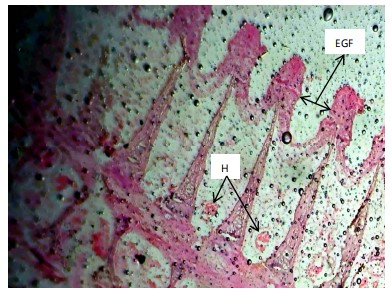
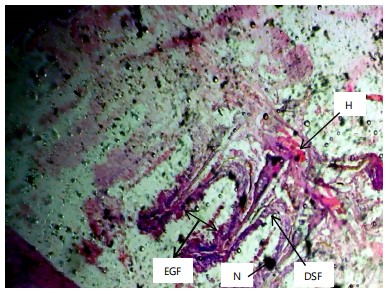
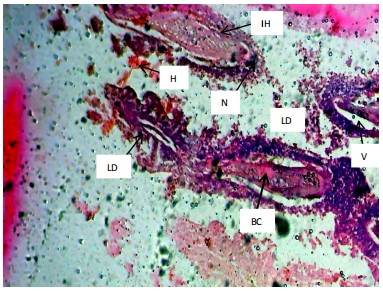
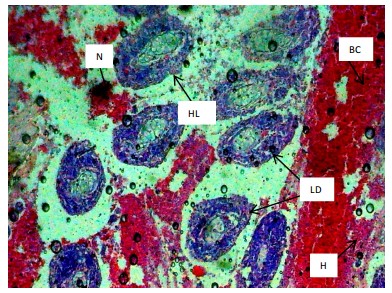
Similarly, the gill filaments (Fig. 8-13) of the control fish showed a normal structure of primary and secondary lamellae. Herbicide exposure caused progressive architectural disruption. Initial damage at 0.59 mg/L included haemorrhage (H) and enlargement of gill filaments (EGF). Higher concentrations (0.88-1.31 mg/L) introduced necrosis (N), distortion and shortening of filaments (DSF), and blood congestion (BC). At the highest concentrations (1.97-2.95 mg/L), the gills exhibited severe lamellar disorganization (LD) and, notably, hyperplasia of the lamellar epithelium (HLE) at 2.95 mg/L, indicating a severe defensive response to the toxicant.
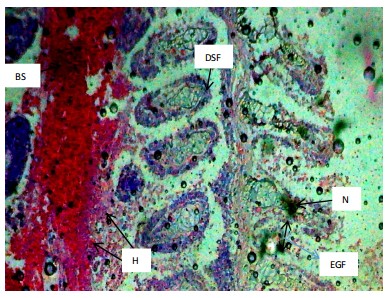
|
|
DISCUSSION
Bispyribac sodium is a widely used herbicide for weed control to enhance agricultural productivity6. However, its application poses risks of food contamination and adverse effects on non-target aquatic species, including invertebrates and vertebrates10. As Clarias gariepinus is a key aquaculture species consumed widely in Nigeria, the potential for Bispyribac sodium residues to accumulate in fish tissues raises significant food safety concerns, potentially affecting human health through dietary exposure6. The present study determined the 96 hrs LC50 of Bispyribac sodium for Clarias gariepinus juveniles as 1.658 mg/L (95% confidence limits: 1.204-2.289 mg/L), indicating moderate toxicity. In contrast, studies on cypermethrin, another pesticide, reported varied LC50 values: 4.17 mg/L for Clarias gariepinus11, 21.4 μg/L for guppy (Poecilia reticulata) over 48 hrs12, and 0.0020-0.0031 μg/L for freshwater prawn (Palaemonetes argentinus) over 24-96 hrs. Additionally, a study by Dey and Saha13 reported higher 96 hrs LC50 values for cypermethrin (0.22-168.07 mg/L) across different species. These discrepancies highlight that herbicide toxicity varies significantly with species, developmental stage, and testing protocols14,15.
|
The observed LC50 of 1.658 mg/L for Clarias gariepinus juveniles indicates that Bispyribac sodium is less toxic to this species compared to carp but more toxic than some cypermethrin LC50 values for Clarias gariepinus. The variation may be attributed to differences in metabolic rates, detoxification mechanisms, or exposure conditions. Clarias gariepinus, known for its resilience to environmental stressors, still exhibited significant toxicity at low concentrations (0.59-2.95 mg/L), with mortality increasing in a concentration-dependent manner (R2 = 0.9552). This aligns with another study16, who noted that herbicide toxicity is both concentration and exposure-dependent.
Behavioural and physiological responses in Clarias gariepinus juveniles exposed to Bispyribac sodium included uncoordinated movement, vertical swimming, gasping, restlessness, loss of equilibrium, skin haemorrhage, and mucus accumulation. These symptoms suggest respiratory impairment and neurological stress, consistent with findings by Collen et al.5 for cypermethrin-exposed prawns. The observed mucus accumulation on gills likely exacerbated respiratory distress, contributing to mortality, particularly at higher concentrations (75% mortality at 2.95 mg/L). Histological changes in liver and gill tissues, such as necrosis, vacuolation, and blood congestion, further indicate systemic toxicity, raising concerns about the quality and safety of fish exposed to Bispyribac sodium for human consumption17.
Physicochemical parameters of the test solutions showed significant changes (p<0.05) in pH (6.15 to 8.05), TDS (327.0 to 358.0 mg/L), EC (654.0 to 716.0 μS/cm), and DO (5.60 to 2.75 mg/L) with increasing Bispyribac sodium concentrations, while temperature remained stable (27.20-27.50°C, P = 0.432). Although these parameters fluctuated, they remained within the tolerance range for Clarias gariepinus as suggested by Arjmandi et al.7. However, the significant decrease in DO (from 2.75 mg/L to 2.95 mg/L) may have amplified toxicity, as low oxygen levels can exacerbate stress in fish, potentially contributing to the observed mortality alongside the direct toxic effects of Bispyribac sodium. The increase in pH, TDS, and EC likely reflects the chemical properties of the herbicide’s sodium-based formulation, which introduces ions into the water. These water quality changes could facilitate herbicide residue uptake in fish tissues, further amplifying food safety risks for consumers18.
The concentration-dependent mortality and water quality changes underscore the ecological risks of Bispyribac sodium in aquatic environments. Synthetic herbicides, including Bispyribac sodium, can cause significant mortality in non-target aquatic organisms and degrade water quality, threatening ecosystem health. The observed liver and gill damage in Clarias gariepinus suggests potential bioaccumulation of Bispyribac sodium, which could compromise the safety of this species as a food source, necessitating stringent monitoring of herbicide residues in aquaculture10. These findings emphasize the need for cautious application of Bispyribac sodium near water bodies and highlight the importance of species-specific toxicity assessments and regulatory measures to ensure food safety in fish consumption.
CONCLUSION
The 96 hrs LC50 of Bispyribac sodium for Clarias gariepinus juveniles was 1.658 mg/L, indicating moderate toxicity. Mortality increased with concentration (R2 = 0.9552), accompanied by stress responses like gasping, uncoordinated movement, and mucus accumulation. Significant changes in pH, TDS, EC, and DO (decreasing to 2.75 mg/L) suggest water quality impacts, with low DO likely amplifying toxicity. Compared to other species, toxicity varies, reflecting species-specific sensitivity. These findings highlight the ecological and food safety risks of Bispyribac sodium, as residues may accumulate in Clarias gariepinus, a widely consumed fish, posing potential health risks to humans. Cautious use near water bodies, further studies on chronic effects, and residue monitoring in aquaculture are essential to ensure ecosystem health and safe fish consumption.
SIGNIFICANCE STATEMENT
This study discovered the acute toxic threshold and water quality disruptions caused by Bispyribac sodium exposure in Clarias gariepinus juveniles, which can be beneficial for environmental risk assessment and aquaculture management. The findings provide quantitative toxicity benchmarks and highlight the sensitivity of this widely cultured species to herbicide contamination. This study will help researchers to uncover the critical areas of herbicide-induced aquatic stress and bioindicator relevance that many researchers were not able to explore. Thus, a new theory on fish-based early warning systems for agrochemical pollution may be arrived at.
REFERENCES
- Blakely, T.J., H.S. Eikaas and J.S. Harding, 2014. The Singscore: A macroinvertebrate biotic index for assessing the health of Singapore’s streams and canals. Raffles Bull. Zool., 62: 540-548.
- Collocott, S.J., L. Vivier and D.P. Cyrus, 2014. Prawn community structure in the subtropical Mfolozi-Msunduzi estuarine system, KwaZulu-Natal, South Africa. Afr. J. Aquat. Sci., 39: 127-140.
- Bonada, N., N. Prat, V.H. Resh and B. Statzner, 2006. Developments in aquatic insect biomonitoring: A comparative analysis of recent approaches. Ann. Rev. Entomol., 51: 495-523.
- Chen, Q.H., W.J. Chen and Z.L. Guo, 2018. Caridean prawn (Crustacea, Decapoda) from Dong'ao Island, Guangdong, China. Zootaxa, 4399: 315-328.
- Collen, B., F. Whitton, E.E. Dyer, J.E.M. Baillie and N. Cumberlidge et al., 2014. Global patterns of freshwater species diversity, threat and endemism. Global Ecol. Biogeogr., 23: 40-51.
- Usmani, K.A and C.O. Knowles, 2001. Toxicity of pyrethroids and effect of synergists to larval and adult Helicoverpa zea, Spodoptera frugiperda and Agrotis ipsilon (Lepidoptera: Noctuidae). J. Econ. Entomol., 94: 868-873.
- Arjmandi, R., M. Tavakol and M. Shayeghi, 2010. Determination of organophosphorus insecticide residues in the rice paddies. Int. J. Environ. Sci. Technol., 7: 175-182.
- Ghazala, S. Mahboob, K.A. Al-Ghanim, S. Sultana, H.F.A. Al-Balawi, T. Sultana and F. Al-Misned, 2014. Acute toxicity II: Effect of organophosphates and carbmates to Catla catla fingerlings. J. Anim. Plant Sci., 24: 1795-1801.
- Yıldırım, Ö., Ü. Acar, R. Tezel, Y. Erden, G. Bilge and S. Yapıcı, 2025. Dose-dependent hemato-biochemical and genotoxic responses of common carp (Cyprinus carpio) to flupyradifurone. Front. Physiol., 16.
- Inyang, I.R., S. Thomas and S.C. Izah, 2016. Activities of electrolytes in kidney and liver of Clarias gariepinus exposed to fluazifop-p-butyl. J. Biotechnol. Res., 2: 68-72.
- Mohamed, H.M.A., W.F.A. Emeish, A. Braeuning and S. Hammad, 2017. Detection of aflatoxin-producing fungi isolated from Nile tilapia and fish feed. EXCLI J., 16: 1308-1318.
- Iheanacho, S.C. and G.E. Odo, 2020. Dietary exposure to polyvinyl chloride microparticles induced oxidative stress and hepatic damage in Clarias gariepinus (Burchell, 1822). Environ. Sci. Pollut. Res., 27: 21159-21173.
- Dey, C. and S.K. Saha, 2014. A comparative study on the acute toxicity bioassay of dimethoate and lambda-cyhalothrin and effects on thyroid hormones of freshwater teleost fish Labeo rohita (Hamilton). Int. J. Environ. Res., 8: 1085-1092.
- Isaac, A.O., O.S. Joshua and A. Jehu, 2017. Behavioural and some physiological assessment of glyphosate and paraquat toxicity to juveniles of African catfish, Clarias gariepinus. Pak. J. Zool., 49: 183-190.
- Burgos-Aceves, M.A., L. Lionetti and C. Faggio, 2019. Multidisciplinary haematology as prognostic device in environmental and xenobiotic stress-induced response in fish. Sci. Total Environ., 670: 1170-1183.
- Rafiq Ullah, A. Zuberi, M. Naeem and Sana Ullah, 2015. Toxicity to hematology and morphology of liver, brain and gills during acute exposure of Mahseer (Tor putitora) to cypermethrin. Int. J. Agric. Biol., 17: 199-204.
- Ezemonye, L. and T.E. Ogbomida, 2010. Histopathological effects of Gammalin 20 on African catfish (Clarias gariepinus). Appl. Environ. Soil Sci., 2010.
- Adeboyejo, O.A., E.O. Clarke and M.O. Olarinmoye, 2011. Organochlorine pesticide residues in water, sediments, fin and shell-fish samples from Lagos Lagoon Complex, Nigeria. Researcher, 3: 38-45.
How to Cite this paper?
APA-7 Style
Kwaghvihi,
O.B., Aende,
A.A., Makeri,
V.A., Akange,
E.T., Ogbonnaya,
M.N. (2026). Acute Toxicity of Bispyribac Sodium on the Survival of African Catfish (Clarias gariepinus) Juveniles and its Implications for Food Safety. Trends in Pharmacology and Toxicology, 2(1), 1-12. https://doi.org/10.21124/tpt.2026.01.12
ACS Style
Kwaghvihi,
O.B.; Aende,
A.A.; Makeri,
V.A.; Akange,
E.T.; Ogbonnaya,
M.N. Acute Toxicity of Bispyribac Sodium on the Survival of African Catfish (Clarias gariepinus) Juveniles and its Implications for Food Safety. Trends Pharm. Toxicol. 2026, 2, 1-12. https://doi.org/10.21124/tpt.2026.01.12
AMA Style
Kwaghvihi
OB, Aende
AA, Makeri
VA, Akange
ET, Ogbonnaya
MN. Acute Toxicity of Bispyribac Sodium on the Survival of African Catfish (Clarias gariepinus) Juveniles and its Implications for Food Safety. Trends in Pharmacology and Toxicology. 2026; 2(1): 1-12. https://doi.org/10.21124/tpt.2026.01.12
Chicago/Turabian Style
Kwaghvihi, Orfega, Benjamin, Aondoahemen Athanasius Aende, Victoria Averem Makeri, Edward Terhemen Akange, and Miracle Nneoma Ogbonnaya.
2026. "Acute Toxicity of Bispyribac Sodium on the Survival of African Catfish (Clarias gariepinus) Juveniles and its Implications for Food Safety" Trends in Pharmacology and Toxicology 2, no. 1: 1-12. https://doi.org/10.21124/tpt.2026.01.12

This work is licensed under a Creative Commons Attribution 4.0 International License.



