Antivenom Potential of Catunaregam nilotica Root-Bark Extract Against Echis ocellatus Venom Toxicity in Albino Rats
| Received 25 Oct, 2025 |
Accepted 23 Jan, 2026 |
Published 31 Mar, 2026 |
Background and Objective: Snakebite envenomation caused by Echis ocellatus (West African carpet viper) remains a major public health issue in Sub-Saharan Africa, contributing significantly to morbidity and mortality. The envenomation commonly results in systemic hemorrhage, coagulation disorders, local tissue necrosis and multiple organ dysfunctions. This study, therefore, investigated the antivenom efficacy of the n-hexane fraction (n-HF) derived from the root-bark of Catunaregam nilotica against E. ocellatus venom-induced pathophysiological alterations in albino rats. Materials and Methods: Acute oral toxicity of the crude methanol extract of C. nilotica root bark was evaluated using Lorke’s method, while the Median Lethal Dose (LD50) of E. ocellatus venom was determined via intraperitoneal administration. Envenomed rats received treatments with the crude extract and its solvent fractions. Standard biochemical procedures were used to determine hepatic and renal function indices, hemostatic parameters (bleeding and clotting times, plasma fibrinogen levels), and lesion diameters. Data were analyzed using one-way ANOVA followed by Tukey’s post hoc test (p<0.05). Results: The crude extract was found to be safe up to 5000 mg/kg body weight, indicating a wide margin of safety. Envenomed rats showed severe pathological effects, including hemorrhage, necrosis, coagulopathy, hepatic and renal impairment, and hemolysis. Administration of both the crude extract and n-HF significantly (p<0.05) improved survival, normalized liver and kidney biomarkers, shortened bleeding and clotting times, reduced hemorrhagic and necrotic lesions, and markedly inhibited hemolysis. Conclusion: The n-hexane fraction of Catunaregam nilotica root bark exhibited potent antivenom activity, effectively mitigating both systemic and local toxic effects of E. ocellatus venom. These findings provide scientific support for the potential development of C. nilotica-based phytotherapeutics as complementary or alternative antivenom agents.
| Copyright © 2026 Sani et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Snakebite envenomation remains a serious but often overlooked public health issue. It primarily affects rural communities in developing countries, where access to healthcare and effective disease surveillance remains limited. The World Health Organization (WHO) classifies snakebite as a neglected tropical disease because of its high rates of death, disability, and economic loss among farming populations who are most at risk1. Globally, around 5.4 mL snakebites occur each year, resulting in about 2.7 mL cases of envenomation and between 81,000 and 138,000 deaths2. Many of those who survive are left with long-term disabilities that affect their livelihoods and quality of life. In Nigeria, Echis ocellatus accounts for more than 90 percent of snakebite deaths, particularly in the northern agricultural belt where most victims are small-scale farmers3. The venom of E. ocellatus is a potent mixture of toxins. Such as Snake-Venom Metalloproteinases (SVMPs), Phospholipase A2 (PLA2), and serine proteases interfere with blood clotting, destroy tissue structures, and can trigger massive bleeding or multi-organ failure4. The standard treatment Anti-Snake Venom (ASV), can neutralize circulating toxins but comes with several challenges. It is expensive, requires cold-chain storage, and is often scarce in rural health facilities. In addition, some patients experience severe allergic reactions, including serum sickness and anaphylaxis5. Unfortunately, ASV also does little to prevent the local tissue damage and necrosis that cause permanent disability among many survivors6. Because of these limitations, there is growing interest in safer, cheaper, and locally available therapeutic options.
Medicinal plants offer a promising alternative since many contain natural compounds with antioxidant, anti-inflammatory, and enzyme-inhibitory properties. Catunaregam nilotica (Rubiaceae); a shrub widely found in the dry regions of Africa. Traditional healers have long used it to treat snakebites, fever, and various infections7. Phytochemical studies of its root bark have identified several bioactive constituents, including flavonoids, alkaloids, tannins, and triterpenoids, all of which have known pharmacological potential8. Although ethnobotanical reports and preliminary laboratory findings suggest that C. nilotica may possess antivenom activity, comprehensive scientific validation is still limited. Therefore, this study investigates the in vivo and in vitro antivenom efficacy of Catunaregam nilotica root-bark extract and its solvent fractions against Echis ocellatus venom, with the goal of identifying plant-based therapeutic candidates that could improve snakebite management in resource-limited settings.
MATERIALS AND METHODS
Plant material collection and identification: Root bark of Catunaregam nilotica was collected in February, 2024 from Gabake Village, Zamfara State, Nigeria. The plant was authenticated by a Taxonomist, and a voucher specimen (KSUSTA/PSB/H/234A) was deposited at the Herbarium of Abdullahi Fodio (Kebbi State) University of Science and Technology, Aliero, Nigeria.
Preparation and fractionation of extract: The air-dried powdered root-bark (500 g) was soaked in 2.5 L of methanol for 72 hrs with intermittent shaking. The filtrate was concentrated at 40°C under reduced pressure to obtain the crude methanol extract of Catunaregam nilotica root bark (CMECNR). The extract was dissolved in water and successively partitioned with n-hexane, ethyl acetate, and n-butanol. Each fraction was dried and stored at 4°C until use9.
Venom source and preparation: Lyophilized Echis ocellatus venom was obtained from Ahmadu Bello University, Zaria, in March 2024. It was dissolved in phosphate-buffered saline (PBS, pH 7.4), aliquoted, and stored at -20°C. Fresh dilutions were prepared for each test.
Experimental animals: Healthy male albino rats (180-220 g) were obtained from the KSUSTA animal facility and maintained under standard laboratory conditions with unrestricted access to feed and water. All procedures followed institutional ethical guidelines for animal care and use.
Acute oral toxicity: The acute oral toxicity of CMECNR was assessed using Lorke’s method with doses ranging from 10 to 5000 mg/kg. Animals were observed for 14 days for behavioral changes or mortality10. The LD50 was calculated as the geometric mean of the highest non-lethal and lowest lethal doses using the formula:
Where:
| a | = | The highest dose at which no death occurred in the second phase | |
| b | = | The lowest dose at which death occurred in the 2nd phase |
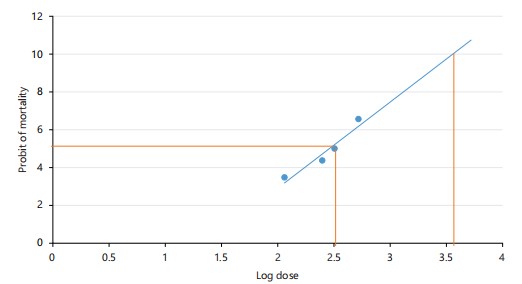
Determination of venom LD50: The LD50 and LD100 of E. ocellatus venom were determined by administering graded intraperitoneal doses to rats (n = 5/group). Mortality after 24 hrs was recorded, and LD50 was estimated using probit analysis11.
In vivo antivenom evaluation of crude extract: Twenty-four rats were divided into six groups (n = 4). The venom control received LD100 venom only, while treatment groups received the venom followed 30 min later by CMECNR (300 or 500 mg/kg, orally) or standard antiserum (1 mL per 0.25 mg venom, i.v.). Control groups received extract or distilled water alone12.
In vivo antivenom evaluation of extract fractions: Twenty-eight rats (n = 4/group) were injected with LD100 venom, followed 30 min later by administration of n-hexane, ethyl acetate, n-butanol, or aqueous fractions (300 mg/kg). Antiserum served as a positive control. Mortality and survival time were recorded for 24 hrs12.
Hepato- and nephro-protective studies: Rats (n = 5/group) were divided into normal, venom control, ASV-treated, and n-hexane fraction-treated (300 mg/kg) groups. After 24 hrs, blood samples were collected for biochemical assays of liver and kidney function using Randox diagnostic kits.
Evaluation of haemostatic parameters: Rats (n = 5/group) were categorized as normal, venom control, ASV-treated, and n-hexane fraction-treated (300 mg/kg). Bleeding time, clotting time, and plasma fibrinogen were assessed 2-4 hrs post-treatment using standard procedures12-14.
Antihaemorrhagic activity: The minimum haemorrhagic dose (MHD) was the lowest venom concentration causing a 10 mm lesion within 6 hrs. Lesion diameters were measured, and percentage inhibition was calculated relative to the venom control12.
Antinecrotic activity: The minimum necrotizing dose (MND) was the venom dose producing a 5 mm lesion after 72 hrs. Lesion sizes were recorded after three days, and inhibition (%) was determined relative to control values12.
Antihaemolytic activity: Erythrocyte suspensions (1%) were incubated with venom (0.1 mg/mL) at 37°C for 30 min, and the absorbance of the supernatant was read at 540 nm. For inhibition assays, venom was pre-incubated with n-hexane fraction (0.5 mg/mL)15. Percentage haemolysis and protection were calculated as follows:
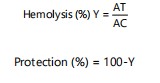 |
Where:
| AT | = | Absorbance of treated sample | |
| AC | = | Absorbance of control |
Statistical analysis: All tests were conducted in triplicate. Data were expressed as Mean±SD and analyzed using one-way ANOVA followed by Tukey’s post hoc test (p<0.05). Statistical analyses were performed with SPSS version 20.
RESULTS
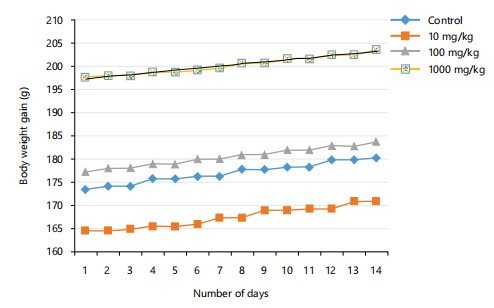
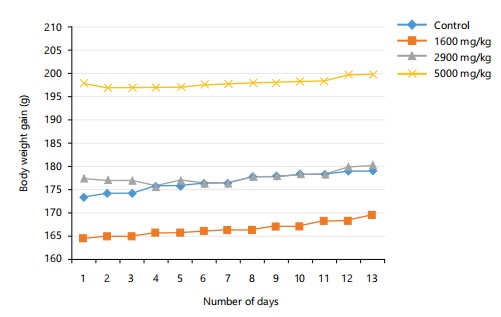
Acute toxicity (LD50): The acute oral toxicity test of the CMECNR revealed no observable toxicity or mortality in rats, even at a dose of 5000 mg/kg as presented in Table 1. Treated animals remained healthy, showing normal behavior and weight gain as shown in Fig. 1 and 2, comparable to the control group.
Lethality of Echis ocellatus venom: Probit analysis revealed that the LD50 and LD100 of Echis ocellatus venom were 0.316 mg/kg body weight and 3.55 mg/kg body weight, respectively. Mortality increases in a dose-dependent manner within 24 hrs of injection, as shown in Table 2 and Fig. 3.
In vivo antivenom activity of crude extract: Rats given the LD100 dose of venom showed complete mortality with a mean survival time of 2.46±0.10 hrs. Treatment with CMECNR significantly (p<0.05) extended survival in a dose-dependent manner, with the 500 mg/kg dose offering 75% protection (MST = 20.11±2.28 hrs), similar to the standard antivenom, which achieved full survival (MST = 24.00±0.00 hrs) (Table 3).
In vivo antivenom activity of extract fractions: The n-hexane fraction (n-HF) of CMECNR showed the most significant antivenom effect (MST = 22.88±2.28 hrs; 75% survival), comparable to ASV (MST = 24.00±0.00 hrs; 100% survival). Ethyl acetate and n-butanol fractions demonstrated moderate antivenom effects, while the aqueous fraction had minimal protection (Table 4).
|
|
|
| Table 1: | Acute lethal effect of crude methanol extract of Catunaregam nilotica root-bark | |||
| Experiment | Dose | Mortality |
| Phase I | 10 | 0/3 |
| 100 | 0/3 | |
| 1000 | 0/3 | |
| Control | Normal saline | 0/3 |
| Phase II | 1600 | 0/1 |
| 2900 | 0/1 | |
| 5000 | 0/1 | |
| Control | Normal saline | 0/1 |
| Table 2: | Probit analysis for determination of venom LD50 and LD100 | |||
| Groups | Average albino rat weight (g) |
Venom dose (mg/kg b.wt) |
Average venom administered (μg/kg b.wt) |
Log dose | No of death/ No of rats used |
Death (%) | Corrected (%)* | Probit of Mortality |
| 1 | 150.4 | - | - | - | 0/4 | 0 | 0 | - |
| 2 | 115.2 | 1 | 115.2 | 2.06 | 0/4 | 0 | *6.25 | 3.45 |
| 3 | 122.2 | 2 | 244.4 | 2.4 | 01-Apr | 25 | 25 | 4.33 |
| 4 | 109.1 | 3 | 327.3 | 2.51 | 02-Apr | 50 | 50 | 5 |
| 5 | 130.5 | 4 | 522 | 2.72 | 04-Apr | 100 | 93.75 | 6.55 |
| Table 3: | Protective effect of CMECNR on survival in Echis ocellatus envenomed rats | |||
| Groups | Treatment | Extract (mg/kg b.wt) |
Venom (mg/kg b.wt) |
Standard antivenin (1 mL/0.25 mg venom) |
Survival/ Total rats |
Survival (%) | Mean survival time (hours) |
| 1 | Control (1% tween 80) | - | 3.55 | - | 04-Apr | 100 | 24.00±0.00c |
| 2 | Venom only | - | 3.55 | - | 0/4 | 0 | 2.46±0.10a |
| 3 | Venom+extract | 300 | 3.55 | - | 01-Apr | 25 | 15.43±2.94b |
| 4 | Venom+extract | 500 | 3.55 | - | 03-Apr | 75 | 20.11±2.28c |
| 5 | Extract only | 500 | 3.55 | - | 04-Apr | 100 | 24.00±0.00c |
| 6 | Venom+ASV | - | 3.55 | 1 mL/0.25 mg venom | 04-Apr | 100 | 24.00+0.00c |
| Mean survival times were presented as Mean±SEM (n = 4), mean survival times carrying different superscripts are significantly (p<0.05) different and -: Not administered | |||||||
| Table 4: | Anti-venom activity of CMECNR fractions on Echis ocellatus venom | |||
| Groups | Treatment | Extract (mg/kg b.wt) |
Venom (mg/kg b.wt) |
Standard antivenin (1 mL/0.25 mg venom) |
Survival/ Total rats |
Survival (%) | Mean survival time (hours) |
| 1 | Control (1% tween 80) | - | 3.55 | - | 04-Apr | 100 | 24.00±0.00c |
| 2 | Venom only | - | 3.55 | - | 0/4 | 0 | 1.40±0.18a |
| 3 | Venom+n-HF | 300 | 3.55 | - | 03-Apr | 75 | 22.88±2.28c |
| 4 | Venom+EAF | 300 | 3.55 | - | 02-Apr | 50 | 14.61±5.42b |
| 5 | Venom+n-BF | 300 | 3.55 | - | 01-Apr | 25 | 11.39±4.41b |
| 6 | Venom+AF | 300 | 3.55 | - | 0/4 | 0 | 6.72±0.57ab |
| 7 | Venom+ASV | - | 3.55 | 1 mL/0.25 mg venom | 04-Apr | 100 | 24.00±0.00c |
| n-HF: n-Hexane fraction, EAF: Ethyl acetate fraction, n-BF: n-Butanol fraction, AF: Aqueous fraction, ASV: Anti-snake venom, Mean survival times were presented as Mean±SEM (n = 4), mean survival times carrying different superscripts are significantly (p<0.05) different and -: Not administered | |||||||
| Table 5: | Effect of Catunaregam nilotica root-bark n-hexane fraction on liver function indices of Echis ocellatus envenomed albino rats | |||
| Normal control | Venom control | Venom+ASV | Venom+n-HF | |
| Total protein (g/L) | 13.34±0.44d | 7.10±0.25a | 8.65±0.25b | 10.06±0.36c |
| Albumin (g/L) | 07.75±0.04d | 05.24±0.10a | 06.69±0.07c | 05.86±0.11b |
| Total Bilirubin (mg/dL) | 00.87±0.05b | 01.93±0.57c | 00.69±0.08a | 00.79±0.06ab |
| Direct Bilirubin (mg/dL) | 00.32±0.03a | 00.72±0.02d | 00.37±0.02b | 00.48±0.01c |
| AST activity (U/L) | 13.55±0.07a | 46.93±0.72c | 18.71±0.11b | 19.53±0.43b |
| ALT activity (U/L) | 29.76±0.44b | 69.87±0.52c | 24.93±0.63a | 30.27±0.50b |
| ALP activity (U/L) | 170.67±0.88b | 240.00±1.73d | 166.67±0.58a | 181.60±1.73c |
| Results are presented as Mean±SEM (n = 4), values carrying different superscripts from the normal control across the row are significantly (p<0.05) different using ANOVA and Duncan multiple range test | ||||
| Table 6: | Effect of Catunaregam nilotica root-bark n-hexane fraction on renal function indices of Echis ocellatus envenomed albino rats | |||
| Normal control | Venom control | Venom+ASV | Venom+n-HF | |
| Urea (mmol/L) | 05.90±0.14a | 07.96±0.14c | 06.19±0.09a | 07.52±0.09b |
| Cretinine (μmol/L) | 99.01±0.82a | 125.00±1.63d | 114.25±2.06c | 105.50±1.73b |
| Uric acid (mmol/L) | 00.49±0.02a | 00.51±0.01a | 00.59±0.02a | 00.47±0.03a |
| Na (mmol/L) | 147.28±0.03d | 146.43±0.06c | 145.57±0.06b | 142.27±0.03a |
| K (mmol/L) | 03.63±0.05a | 4.51±0.03d | 04.30±0.02c | 04.20±0.02b |
| Cl– (mmol/L) | 104.33±33a | 104.50±0.07b | 104.54±0.01b | 104.71±0.03c |
| Results are presented as Mean±SEM (n = 4), values carrying different superscripts from the normal control across the row are significantly (p<0.05) different using ANOVA and Duncan multiple range test | ||||
Effect on hepatic function: Envenomation caused a significant (p<0.05) elevation in hepatic enzymes (AST, ALT, ALP) and bilirubin levels, alongside a decline in total protein and albumin. Treatment with n-HF effectively restored these indices toward normal values, comparable to ASV (Table 5).
Effect on renal function: Venom administration led to elevated urea, creatinine, and potassium concentrations. Treatment with n-HF significantly (p<0.05) decreased creatinine and potassium levels more effectively than ASV, while both treatments normalized urea and sodium concentrations. Uric acid and chloride levels remained unchanged (Table 6).
| Table 7: | Effects of Catunaregam nilotica root-bark n-hexane fraction on hemostatic parameters in Echis ocellatus-envenomed rats | |||
| Treatment group | Bleeding time (s) | Clotting time (s) | Defibrinogenating effect |
| Normal control | 63.75±0.75a | 93.75±1.55a | 63.75±1.50a |
| Venom control | 128.00±1.08d | 195.50±1.66c | NC |
| Venom+ASV | 79.50±1.70b | 115.00±1.78b | 120.25±1.38b |
| Venom+n-HF | 90.75±2.39c | 119.50±0.87b | 124.00±1.67b |
| Results are presented as Mean±SEM (n = 4), values with different superscripts across rows differ significantly (p<0.05) and NC: No clotting | |||
| Table 8: | Effects of Catunaregam nilotica root-bark n-hexane fraction on hemorrhagic and necrotic lesions in Echis ocellatus-envenomed rats | |||
| Treatment group | Hemorrhagic lesion (mm) | Necrotic lesion (mm) |
| Normal control | 0.00±0.00a | 0.00±0.00a |
| Venom control | 11.05±0.01d | 16.63±0.94c |
| Venom+ASV | 2.33±0.08b | 5.78±0.88b |
| Venom+n-HF | 4.23±0.20c | 7.22±0.60b |
| Results are presented as Mean±SEM (n = 4), Different superscripts indicate significant differences (p<0.05) | ||
| Table 9: | Effects of Catunaregam nilotica root-bark n-hexane fraction on hemolysis in Echis ocellatus-envenomed rats | |||
| Treatment group | Hemolysis (%) |
| Normal control | 2.60±0.16a |
| Venom control | 90.70±2.18d |
| Venom+ASV | 25.18±0.71b |
| Venom+n-HF | 29.69±1.61c |
| Results are presented as Mean±SEM (n = 4), values with different superscripts differ significantly (p<0.05) | |
Effects on hemostatic parameters: Echis ocellatus envenomation significantly prolonged bleeding (128.00±1.08 sec) and clotting (195.50±1.66 sec) times, indicating coagulopathy. Administration of n-HF markedly shortened these times (90.75±2.39 and 119.50±0.87 sec, respectively), comparable to ASV-treated rats. The defibrinogenating effect, absent in venom-only rats, was restored by both treatments (Table 7).
Antihaemorrhagic and antinecrotic effects: Venom injection produced severe hemorrhagic (11.05±0.01 mm) and necrotic (16.63±0.94 mm) lesions. Treatment with n-HF significantly (p<0.05) reduced lesion diameters (4.23±0.20 and 7.22±0.60 mm, respectively), comparable to ASV (2.33±0.08 mm and 5.78±0.88 mm) (Table 8).
Antihaemolytic activity: Venom exposure caused extensive hemolysis (90.70±2.18%). Treatment with n-HF markedly reduced hemolysis to 29.69±1.61%, comparable to ASV (25.18±0.71%). This indicates strong erythroprotective and membrane-stabilizing potential of the n-HF fraction (Table 9).
DISCUSSION
The crude methanol extract of Catunaregam nilotica root bark showed no signs of toxicity or mortality up to 5000 mg/kg, indicating a broad safety margin and classifying it as practically non-toxic according to OECD (2001) standards. This observation agrees with findings from other Rubiaceae species16. The LD50 and LD100 values of Echis ocellatus venom were consistent with previous reports17,18. Minor differences in venom potency reported may reflect geographical and ecological variations in venom composition19. In vivo studies revealed that E. ocellatus envenomation caused typical viperid symptoms, hemorrhage, necrosis, coagulopathy, and organ dysfunction. Treatment with the crude extract of C. nilotica improved survival and reduced tissue injury, suggesting the presence of antivenom compounds. Among the solvent fractions, the n-hexane fraction (n-HF) was the most effective, similar to reports on Azadirachta indica and Albizia chevalieri9,19. Its strong activity may be linked to non-polar terpenoids, steroids, and alkaloids that chelate metal ions, inhibit venom enzymes, and reduce oxidative stress20. Envenomed rats exhibited liver and kidney damage, evidenced by increased transaminases, urea, creatinine, and electrolyte imbalance consistent with SVMP- and PLA2-mediated cytotoxicity21. Treatment with n-HF normalized these iochemical parameters, indicating strong hepatoprotective and nephroprotective effects rarely observed with standard antivenoms22,23. Similar recovery has been reported with plant-based antivenoms such as Moringa oleifera, Annona senegalensis, and Andrographis paniculata24-27. The presence of flavonoids, tannins, and terpenoids in n-HF supports its antioxidant, anti-inflammatory, and protease-inhibitory mechanisms3,6. Venom exposure also caused hemostatic disturbances, including prolonged clotting time, defibrinogenation, and local hemorrhage. The n-HF treatment restored normal coagulation, reduced lesion size, and prevented fibrinogen depletion, likely through inhibition of zinc-dependent SVMPs responsible for vascular and extracellular matrix damage28,29. Its suppression of hemolysis further suggests Phospholipase A2 inhibition, preventing erythrocyte lysis and lipid peroxidation30-33.
CONCLUSION
The n-hexane fraction of Catunaregam nilotica root bark demonstrated potent antivenom activity against Echis ocellatus venom by effectively attenuating both systemic and local toxic effects. Treatment significantly improved survival, normalized hepatic and renal biomarkers, restored hemostatic balance, and reduced venom-induced hemorrhagic and necrotic lesions. These findings substantiate the therapeutic relevance of C. nilotica and highlight its potential as a complementary phytotherapeutic agent in snakebite management.
SIGNIFICANCE STATEMENT
Snakebite envenomation by Echis ocellatus remains a major cause of morbidity and mortality in Sub-Saharan Africa. The present findings confirm that the n-hexane fraction of Catunaregam nilotica root bark effectively counteracts venom-induced hemorrhage, necrosis, hemolysis, and organ damage. Unlike conventional antivenoms that primarily neutralize systemic toxins, this plant-derived fraction offers protection against both local and systemic pathologies. The study provides scientific validation for its traditional use and supports its potential development as an affordable, complementary antivenom therapy.
REFERENCES
- Gutiérrez, J.M., J.J. Calvete, A.G. Habib, R.A. Harrison, D.J. Williams and D.A. Warrell, 2017. Snakebite envenoming. Nat. Rev. Dis. Primers, 3. https://doi.org/10.1038/nrdp.2017.63
- Adrião, A.A.X., A.O. dos Santos, E.J.S.P. de Lima, J.B. Maciel and W.H.P. Paz et al., 2022. Plant-derived toxin inhibitors as potential candidates to complement antivenom treatment in snakebite envenomations. Front. Immunol., 13.
- Habib, A.G., B.M. Musa, G. Iliyasu, M. Hamza, A. Kuznik and J.P. Chippaux, 2020. Challenges and prospects of snake antivenom supply in Sub-Saharan Africa. PLoS Negl. Trop. Dis., 14.
- Gutiérrez, J.M., L.O. Albulescu, R.H. Clare, N.R. Casewell, T.M. Abd El-Aziz, T. Escalante and A. Rucavado, 2021. The search for natural and synthetic inhibitors that would complement antivenoms as therapeutics for snakebite envenoming. Toxins, 13.
- Bando, D.C., E.O. Ogu, I. Nuhu, A.D. Suleiman and H.N. Tubasen et al., 2020. Anti-hemorrhagic activity of wild custard apple (Annona senegalensis) ethanolic leaf extract on spitting cobra (Naja negricollis) metalloprotease. Am. Sci. Res. J. Eng. Technol. Sci., 70: 77-89.
- Liaqat, A., T.H. Mallhi, Y.H. Khan, A. Khokhar, S. Chaman and M. Ali, 2022. Anti-snake venom property of medicinal plants: A comprehensive review of literature. Braz. J. Pharm. Sci., 58.
- Sani, I., A.N. Ukwuani-Kwaja, A. Zubairu, I.M. Fakai, F. Bello and H.A. Sani, 2024. Antivenom potentials of some local medicinal plants against Naja nigricollis associated envenoming in North-Western Nigeria. Pharmacologia, 15: 15-22.
- Salihu, M., L.G. Hassan, U.Z. Faruq and A.J. Yusuf, 2024. Deciphering the interactions of scopoletin and scopolin from Catunaregam nilotica roots against Naja nigricollis phospholipase A2 enzyme. Toxicon, 243.
- Mathias, S.N., K. Abubakar, N. October, M.S. Abubakar and H.E. Mshelia, 2016. Anti-venom potentials of friedelin isolated from hexane extract fraction of Albizia chevalieri Hams (Mimosaceae). Ife J. Sci., 18: 473-482.
- Lorke, D., 1983. A new approach to practical acute toxicity testing. Arch. Toxicol., 54: 275-287.
- Broad, A.J., S.K. Sutherland and A.R. Coulter, 1979. The lethality in mice of dangerous Australian and other snake venom. Toxicon, 17: 661-664.
- Theakston, R.D.G. and H.A. Reid, 1983. Development of simple standard assay procedures for the characterization of snake venoms. Bull. World Health Organ., 61: 949-956.
- Mukherjee, A.K., 2012. Green medicine as a harmonizing tool to antivenom therapy for the clinical management of snakebite: The road ahead. Indian J. Med. Res., 136: 10-12.
- Sani, I., R.A. Umar, S.W. Hassan, U.Z. Faruq, F. Bello and A. Abdulhamid, 2020. Inhibition of snake venom enzymes and antivenom adjuvant effects of Azadirachta indica A. Juss. (Meliaceae) leaf extracts. Eur. J. Med. Plants, 31: 114-128.
- Ekor, M., 2014. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol., 4.
- Mors, W.B., M.C. do Nascimento, B.M.R. Pereira and N.A. Pereira, 2000. Plant natural products active against snake bite-the molecular approach. Phytochemistry, 55: 627-642.
- Oliveira, A.L., M.F. Viegas, S.L. da Silva, A.M. Soares, M.J. Ramos and P.A. Fernandes, 2022. The chemistry of snake venom and its medicinal potential. Nat. Rev. Chem., 6: 451-469.
- Dingwoke, E.J., F.A. Adamude, A. Salihu, M.S. Abubakar and A.B. Sallau, 2024. Toxicological analyses of the venoms of Nigerian vipers Echis ocellatus and Bitis arietans. Trop. Med. Health, 52.
- Sani, I., R.A. Umar, S.W. Hassan, U.Z. Faruq and F. Bello, 2020. Isolation of antisnake venom agents from Azadirachta indica (A. Juss) leaf extracts. Acad. J. Chem., 5: 10-16.
- Ajisebiola, B.S., B.P. Durodola, A.R.K. Mustapha, J.O. Oladele and A.O. Adeyi, 2024. Cardio-nephrotoxicity mediated by Echis ocellatus venom and its amelioration through kaempferol’s suppressive effect on oxidative stress, inflammation, and apoptosis expression. Clin. Phytosci., 10.
- Liang, X., D. Xing, Z. Zhuang, H. Feng and S. Li et al., 2025. Prognostic value of red blood cell width distribution-to-platelet ratio in patients with snakebite-associated multiple organ dysfunction syndrome: A retrospective observational study. J. Inflammation Res., 18: 5281-5291.
- Alirol, E., S.K. Sharma, H.S. Bawaskar, U. Kuch and F. Chappuis, 2010. Snake bite in South Asia: A review. PLoS Negl. Trop. Dis., 4.
- Fu, K., J. Zhao, L. Zhong, H. Xu, X. Yu, X. Bi and C. Huang, 2024. Dual therapy with phospholipase and metalloproteinase inhibitors from Sinonatrix annularis alleviated acute kidney and liver injury caused by multiple snake venoms. Biomed. Pharmacother., 177.
- Adeyi, A.O., S.O. Adeyemi, E.O.P. Effiong, B.S. Ajisebiola, O.E. Adeyi and A.S. James, 2021. Moringa oleifera extract extenuates Echis ocellatus venom-induced toxicities, histopathological impairments and inflammation via enhancement of Nrf2 expression in rats. Pathophysiology, 28: 98-115.
- Adzu, B., M.S. Abubakar, K.S. Izebe, D.D. Akumka and K.S. Gamaniel, 2005. Effect of Annona senegalensis rootbark extracts on Naja nigricotlis nigricotlis venom in rats. J. Ethnopharmacol., 96: 507-513.
- Sani, I., R.A. Umar, S.W. Hassan, U.Z. Faruq, F. Bello, H. Aminu and A. Sulaiman, 2020. Hepatoprotective effect of Azadirachta indica leaf fractionated extracts against snake venom toxicity on albino rats. Saudi J. Biomed. Res., 5: 112-117.
- Nayak, A.G., N. Kumar, S. Shenoy and M. Roche, 2020. Anti-snake venom and methanolic extract of Andrographis paniculata: A multipronged strategy to neutralize Naja naja venom acetylcholinesterase and hyaluronidase. 3 Biotech, 10.
- Mourao, V.B., G.M. Giraldi, L.M.G. Neves, F.O. de Gaspari de Gaspi and R.A.F. Rodrigues et al., 2014. Anti-hemorrhagic effect of hydro-alcoholic extract of the leaves of Mikania glomerata in lesions induced by Bothrops jararaca venom in rats. Acta Cirúrgica Bras., 29: 30-37.
- Nguyen, D.D., N.H.A. Lam and D.B. Nguyen, 2025. Severe and prolonged hypofibrinogenemia following Protobothrops cornutus snake bite: A case report from Vietnam. Toxicon, 266.
- Salvador, G.H.M., A.A.S. Gomes, W. Bryan-Quirós, J. Fernández and M.R. Lewin et al., 2019. Structural basis for phospholipase A2-like toxin inhibition by the synthetic compound Varespladib (LY315920). Sci. Rep., 9.
- Zhao, J., G. Liu, X. Shi and C. Huang, 2025. Combination of rhamnetin and RXP03 mitigates venom-induced toxicity in murine models: Preclinical insights into dual-target antivenom therapy. Toxins, 17.
- Enechi, O.C., E.S. Okeke, O.E. Awoh, C.O. Okoye and C.K. Odo, 2021. Inhibition of phospholipase A2, platelet aggregation and egg albumin induced rat paw oedema as anti-inflammatory effect of Peltophorun pterocarpus stem-bark. Clin. Phytosci., 7.
- Abiola, J., O. Aiyelaagbe, A. Adeyi, B. Ajisebiola and S. König, 2025. Strophanthus sarmentosus extracts and the strophanthus cardenolide ouabain inhibit snake venom proteases from Echis ocellatus. Molecules, 30.
How to Cite this paper?
APA-7 Style
Sani,
I., Ukwuani-Kwaja,
A.N., Kankara,
A.I., Bello,
F., Hassan Fakai,
H.J., Muhammad,
H., Abdulrauf,
M.S. (2026). Antivenom Potential of Catunaregam nilotica Root-Bark Extract Against Echis ocellatus Venom Toxicity in Albino Rats. Trends in Pharmacology and Toxicology, 2(1), 13-22. https://doi.org/10.21124/tpt.2026.13.22
ACS Style
Sani,
I.; Ukwuani-Kwaja,
A.N.; Kankara,
A.I.; Bello,
F.; Hassan Fakai,
H.J.; Muhammad,
H.; Abdulrauf,
M.S. Antivenom Potential of Catunaregam nilotica Root-Bark Extract Against Echis ocellatus Venom Toxicity in Albino Rats. Trends Pharm. Toxicol. 2026, 2, 13-22. https://doi.org/10.21124/tpt.2026.13.22
AMA Style
Sani
I, Ukwuani-Kwaja
AN, Kankara
AI, Bello
F, Hassan Fakai
HJ, Muhammad
H, Abdulrauf
MS. Antivenom Potential of Catunaregam nilotica Root-Bark Extract Against Echis ocellatus Venom Toxicity in Albino Rats. Trends in Pharmacology and Toxicology. 2026; 2(1): 13-22. https://doi.org/10.21124/tpt.2026.13.22
Chicago/Turabian Style
Sani, Ibrahim, Angela N. Ukwuani-Kwaja, Aliyu Idris Kankara, Fatima Bello, Habiba Joy Hassan Fakai, Hashimu Muhammad, and Muhammad Shafi’u Abdulrauf.
2026. "Antivenom Potential of Catunaregam nilotica Root-Bark Extract Against Echis ocellatus Venom Toxicity in Albino Rats" Trends in Pharmacology and Toxicology 2, no. 1: 13-22. https://doi.org/10.21124/tpt.2026.13.22

This work is licensed under a Creative Commons Attribution 4.0 International License.




