Chemotherapeutic Potential of a Tri-Herbal Formulation Against NMU-Induced Mammary Tumors via Histological Remodeling and Genoprotection
| Received 31 Oct, 2025 |
Accepted 27 Mar, 2026 |
Published 31 Mar, 2026 |
Background and Objective: Herbal bioactives from Nigella sativa (black cumin), Boswellia sacra (frankincense), and Carica papaya (papaya leaf) possess potent antioxidant and anti-inflammatory activities. This study evaluated the histopathological and genotoxic effects of a tri-herbal formulation combining these botanicals in N-nitrosomethylurea (NMU)-induced mammary carcinogenesis in mice. Materials and Methods: Fifty female mice were randomly divided into 5 groups (n = 10): Control, NMU+cisplatin (2 mg/kg/week), NMU+Tri-herbal low dose (100 mg/kg/day), NMU+tri-herbal mid dose (200 mg/kg/day), and NMU+Tri-herbal high dose (400 mg/kg/day). Mammary tumours were induced via intraperitoneal NMU injections (50 mg/kg) administered weekly for three weeks. One week after the final NMU dose, treatments were given orally for eight weeks. Body weight and tumour growth were monitored weekly. After euthanasia, mammary and visceral tissues were examined histologically (H&E), and DNA damage in blood and tumour cells was quantified using the alkaline comet assay. All quantitative data were analysed using one-way analysis of variance followed by Tukey’s post hoc test, and differences were considered statistically significant at p<0.05. Results: Tri-herbal-treated groups showed significant reductions in tumour volume and improved body weight relative to NMU controls (p<0.05). Histopathology revealed reduced cellular proliferation, increased apoptotic activity, and near-normal glandular restoration. Comet assay indices (% tail DNA, tail moment) indicated markedly decreased genotoxicity compared with NMU+cisplatin, suggesting enhanced genomic stability. Conclusion: The tri-herbal combination of N. sativa, B. sacra, and C. papaya exhibits robust chemopreventive potential against NMU-induced mammary tumours through antioxidative, anti-inflammatory, and DNA-protective mechanisms, supporting its promise as a complementary phytotherapeutic in breast cancer management.
| Copyright © 2026 Sowunmi et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Carcinogenesis is a complex, multistep process driven by oxidative stress, DNA damage, and chronic inflammation1,2. The N-nitrosomethylurea (NMU) model replicates this process by inducing alkylation of DNA bases, leading to mutations and genomic instability in mammary epithelial cells. Continuous exposure to reactive oxygen species (ROS) further amplifies this damage by disrupting redox balance and promoting lipid peroxidation3. Over time, this results in loss of cell-cycle control, oncogene activation, and tumour development4,5.
The cellular response to oxidative stress involves two key regulatory proteins: Nuclear factor erythroid 2-related factor 2 (Nrf2) and the tumour suppressor p53. The Nrf2 maintains redox homeostasis by activating the transcription of antioxidant enzymes such as glutathione peroxidase and superoxide dismutase6. The p53 detects DNA damage and triggers repair or apoptosis depending on the extent of injury. A functional balance between these two pathways determines whether a cell survives or undergoes programmed death7. In cancer cells, this balance is disrupted, and persistent oxidative stress suppresses Nrf2 signalling and impairs p53 activity, leading to genomic instability and malignant progression8,9.
Natural compounds with antioxidant and anti-inflammatory properties can restore this balance. Thymoquinone, a bioactive component of Nigella sativa, activates Nrf2 signalling while enhancing p53-dependent apoptosis in tumour cells10,11. It scavenges ROS and limits lipid peroxidation, which protects mitochondrial membranes and maintains energy balance. Studies have shown that thymoquinone-treated breast cancer cells exhibit reduced oxidative DNA damage and increased expression of DNA repair genes12.
Boswellic acids from Boswellia sacra target inflammatory mediators by inhibiting 5-lipoxygenase and NF-κB activation, both of which drive tumour-associated inflammation13,14. This action decreases the production of pro-inflammatory cytokines and nitric oxide, reducing oxidative stress in the tumour microenvironment. Boswellic acids also induce apoptosis through mitochondrial depolarization and activation of caspase-3, leading to selective elimination of malignant cells while sparing healthy tissues15.
The third component, Carica papaya leaf extract, contributes polyphenols, flavonoids, and the cysteine protease papain, which improve antioxidant defence and immune regulation15. In animal models, papaya extract reduces lipid peroxidation and restores hepatic and renal enzyme profiles after chemical insult. It also enhances glutathione levels and maintains DNA integrity by reducing single-strand breaks16,17.
Together, these three botanicals provide complementary protection. N. sativa supports redox control and DNA repair; B. sacra modulates inflammation and apoptosis; and C. papaya improves detoxification and organ recovery. Their combination is expected to strengthen the Nrf2-p53 regulatory axis, suppress NF-κB-mediated inflammation, and reduce mitochondrial oxidative injury. The result is enhanced DNA stability, controlled cell proliferation, and improved tolerance to oxidative and chemotherapeutic stress18.
Previous studies have highlighted the value of combining phytochemicals to achieve synergistic effects against cancer progression19. Esmaeili et al.12 reported that multi-component herbal formulations modulate apoptosis-related gene expression and reduce genotoxicity in carcinogen-exposed models. Similarly, Dubey and Sharma20 demonstrated that plant-derived antioxidants can restore mitochondrial function and reverse oxidative stress in mammary tissues.
This mechanistic framework provides a scientific basis for evaluating the tri-herbal combination of Nigella sativa, Boswellia sacra, and Carica papaya. Targeting multiple molecular pathways, the formulation has potential to protect genomic integrity, restore tissue architecture, and improve systemic resilience in NMU-induced breast cancer models.
MATERIALS AND METHODS
Study area: The study was carried out at the D. K. Olukoya Central Research and Reference Laboratories, University of Lagos, Lagos, Nigeria. All experimental work, animal handling, and histopathological procedures were conducted within the laboratory-controlled animal research unit over 12 months from April 2020 to March 2021.
Experimental animals: Fifty healthy female mice (8-10 weeks old, 18-25 g) were obtained from an accredited breeding facility. Animals were housed in polypropylene cages (10 mice per group) under standard laboratory conditions (temperature 25±2°C; relative humidity 50-60%; 12 hrs light/dark cycle) with free access to standard chow and water. Animals were acclimatized for one week before experimentation. All procedures conformed to the Guide for the Care and Use of Laboratory Animals (National Research Council, 2011) and were approved by the Institutional Animal Care and Use Committee (protocol UNILAG-CBG/2019/029).
Phytochemical characterization of Tri-Herbal Formulation: The Tri-Herbal Formula (THF) was prepared by mixing ethanolic extracts of Nigella sativa seeds, Boswellia sacra resin, and Carica papaya leaves in a 1:1:1 ratio (w/w). The extract mixture was subjected to preliminary phytochemical screening and chromatographic profiling. Quantitative analysis was performed using high-performance liquid chromatography (HPLC; Agilent 1260 Infinity) with a C18 reverse-phase column and UV detection at 254 nm. Standard curves were generated using thymoquinone, 11-keto-β-boswellic acid, and quercetin as marker compounds. Results were expressed as mg of marker compound per g of dry extract. The extract yield and batch-to-batch consistency were verified before use.
Induction of mammary tumours: Mammary carcinogenesis was induced following established NMU protocols4. N-Nitrosomethylurea (NMU; Sigma-Aldrich) was prepared freshly in acidified saline (0.9% NaCl, pH ≈ 4, adjusted with acetic acid) and injected intraperitoneally at 50 mg/kg body weight once weekly for three consecutive weeks. Handling of NMU was performed under a fume hood with appropriate protective equipment due to its carcinogenicity21. Animals were observed for tumour latency and progression from week 4 onward.
Experimental design and treatment regimen: After successful tumour induction, mice were randomly assigned to five experimental groups (n = 10 per group). Table 1.
Monitoring of physiological parameters: Body weights and food intake were recorded weekly throughout the study. Tumour development was assessed by manual palpation twice weekly. Tumour length (L) and width (W) were measured with Vernier callipers, and tumour volume (V) was calculated using the ellipsoid formula22:
| Table 1: | Experimental grouping and treatment schedule | |||
| Group | Treatment | Description |
| I | Control | Vehicle only |
| II | NMU+Cisplatin | NMU+cisplatin 2 mg/kg i.p. once per week |
| III | NMU+Tri-herbal Low | NMU+tri-herbal 100 mg/kg/day p.o. |
| IV | NMU+Tri-herbal Mid | NMU+tri-herbal 200 mg/kg/day p.o. |
| V | NMU+Tri-herbal High | NMU+tri-herbal 400 mg/kg/day p.o. |
| Treatment commenced one week after the final NMU injection and continued for eight weeks. Doses were chosen based on preliminary acute toxicity and tolerability studies (LD50>2000 mg/kg). Oral gavage was performed daily at 09:00-10:00 hrs | ||
Clinical observations for distress, grooming behaviour, and posture were noted daily. Humane endpoints included tumour diameter >1.5 cm, ulceration, or >20% body-weight loss.
Tissue collection: At the end of the 8-week treatment period, animals were fasted overnight and euthanized under ketamine/xylazine anaesthesia (100/10 mg/kg i.p.). Blood was collected by cardiac puncture into EDTA tubes for the comet assay. Tumour tissue and major organs (liver, kidney, heart, and lung) were excised, weighed, and divided for histopathological and genotoxic analyses. Organs for histology were fixed immediately in 10% neutral-buffered formalin.
Histopathological evaluation: At the end of the experimental period, mammary glands and visceral organs (liver, kidney) were excised, rinsed in cold saline, and fixed in 10% neutral-buffered formalin for 48 hrs. Fixed tissues were processed through graded alcohols, cleared in xylene, and embedded in paraffin wax. Serial sections (5 μm thick) were cut using a rotary microtome and stained with Haematoxylin and Eosin (H&E).
Slides were examined under a light microscope (Leica DM500) at magnifications of ×100 and ×400. Morphological alterations were scored semi-quantitatively on a scale of 0-3 (0 = none, 1 = mild, 2 = moderate, 3 = severe) for necrosis, inflammation, and hyperplasia by a board-certified pathologist blinded to the treatment groups. Representative micrographs were captured using a digital imaging system (Leica ICC50 W).
Genotoxicity assessment-alkaline comet assay: The DNA strand breaks in mammary epithelial cells were evaluated using the alkaline comet assay as previously described by Singh et al.23 with minor modifications to ensure standardization. Briefly, freshly excised mammary tissues were minced in ice-cold phosphate-buffered saline (PBS, pH 7.4) and filtered through a 40 μm nylon mesh to obtain a single-cell suspension. Cell viability (> 85%) was confirmed by trypan blue exclusion.
For slide preparation, pre-cleaned microscope slides were coated with 1% normal-melting-point agarose and allowed to dry. Approximately 10 μL of the cell suspension (1×104 cells/mL) was mixed with 90 μL of 0.5% low-melting-point agarose at 37°C, layered onto the base gel, and covered with a coverslip to solidify at 4°C for 10 min. Slides were then immersed in freshly prepared lysis buffer (2.5 M NaCl, 100 mM EDTA, 10 mM Tris, 1% Triton X-100, and 10% DMSO, pH 10) at 4°C for 1 h to remove cellular membranes and proteins.
Following lysis, slides were placed in electrophoresis buffer (300 mM NaOH, 1 mM EDTA, pH>13) for 20 min at 4°C to allow DNA unwinding, and electrophoresed at 25 V (0.8 V/cm) and 300 mA for 20 min in the dark. Slides were then neutralized in 0.4 M Tris buffer (pH 7.5) for 10 min and dehydrated with absolute ethanol.
The DNA was stained with ethidium bromide (2 μg/mL) and examined under a fluorescence microscope (Olympus BX51, 40× objective) equipped with a 515-560 nm excitation filter. A minimum of 50 nuclei per animal (three slides per animal) were scored using OpenComet v1.3 software (ImageJ plugin). Parameters analysed included tail length (μm), % tail DNA, and tail moment. Scoring was performed by two independent observers blinded to the treatment groups. Data were expressed as median ± interquartile range (IQR), and inter-observer agreement exceeded 0.90 (Cohen’s κ coefficient).
Biochemical and haematological evaluation: Serum was separated for the determination of Alanine Aminotransferase (ALT), Aspartate Aminotransferase (AST), creatinine, and urea using commercial diagnostic kits (Randox Laboratories). Haematological indices (RBC, WBC, Hb, platelets) were analysed using an automated cell counter to assess systemic toxicity.
Statistical analysis: Data were analysed using GraphPad Prism version 10.0 (GraphPad Software, USA) and Microsoft Excel Package. Normality was assessed using the Shapiro-Wilk test and homogeneity of variances with Levene’s test24. Normally distributed datasets were analysed by one-way ANOVA followed by Tukey’s post-hoc multiple-comparison test, while non-normal datasets (comet parameters) were evaluated using the Kruskal-Wallis test with Dunn’s correction. Results are expressed as mean±SEM for parametric data or median±IQR for non-parametric data. Statistical significance was accepted at p<0.05. In addition, effect sizes (η2 for ANOVA; r for Kruskal-Wallis) and 95% confidence intervals were computed to estimate the magnitude of differences between groups.
Ethical approval and animal welfare: All experimental procedures involving animals were conducted in accordance with the ethical principles for laboratory animal use and care as outlined in the National Institutes of Health Guide for the Care and Use of Laboratory Animals (8th edition, 2011) and the ARRIVE 2.0 guidelines. The study protocol was reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of the Department of Cell Biology and Genetics, University of Lagos, Nigeria.
RESULTS
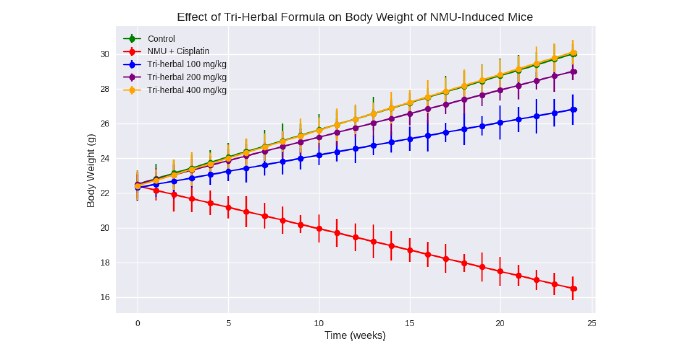
Body weight: Over the six-month experimental period, the physical condition and mean body weight of mice were closely monitored as indicators of systemic toxicity and recovery (Table 2). The untreated control group (A) maintained steady weight gain and normal grooming behaviour. Conversely, NMU+cisplatin-treated mice (Group B) exhibited marked weight loss, poor coat condition, and decreased locomotor activity, reflecting both carcinogenic and chemotherapeutic stress.
Administration of the Tri-herbal formulation composed of Nigella sativa, Boswellia sacra, and Carica papaya mitigated these adverse changes in a dose-dependent pattern. By week 24, the mid- and high-dose groups (D = 200 mg/kg; E = 400 mg/kg) demonstrated significant recovery in body weight compared with the NMU+cisplatin group (p<0.05). The restoration of normal appetite and mobility suggests systemic improvement associated with phytochemical antioxidant and anti-inflammatory activity Fig. 1.
|
|
| Table 2: | Effect of tri-herbal formula on body weight of NMU-Induced mice | |||
| Week | Control (A) | NMU+Cisplatin (B) | Tri-herbal 100 mg/kg (C) | Tri-herbal 200 mg/kg (D) | Tri-herbal 400 mg/kg (E) |
| 0 | 22.5±0.4 | 22.4±0.3 | 22.3±0.3 | 22.5±0.4 | 22.4±0.4 |
| 4 | 24.1±0.6 | 21.0±0.5 | 22.8±0.4 | 23.5±0.5 | 23.8±0.5 |
| 8 | 25.8±0.7 | 19.3±0.6 | 23.6±0.5 | 24.8±0.6 | 25.2±0.6 |
| 12 | 27.0±0.8 | 18.1±0.5 | 24.4±0.6 | 25.9±0.5 | 26.5±0.6 |
| 16 | 28.3±0.7 | 17.5±0.4 | 25.3±0.5 | 27.1±0.5 | 27.9±0.5 |
| 20 | 29.1±0.6 | 17.0±0.5 | 26.0±0.4 | 28.2±0.6 | 29.0±0.5 |
| 24 | 30.0±0.6 | 16.5±0.4 | 26.8±0.5 | 29.0±0.5 | 30.1±0.4 |
| Values are presented as Mean±Standard Deviation (SD) for each group (n = 10). Statistical analysis was performed using one-way ANOVA followed by Tukey’s post hoc test. Differences were considered significant at p<0.05 compared with NMU+cisplatin group | |||||
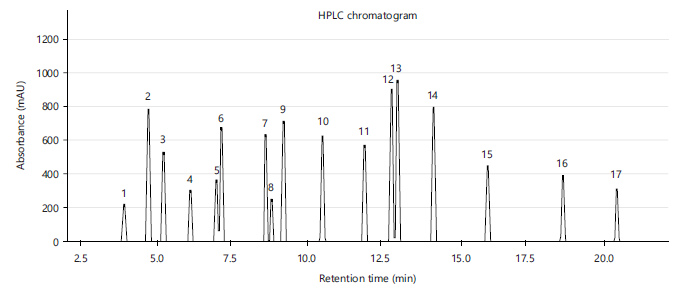
HPLC characterization of the Tri-herbal formula: The HPLC chromatographic profiling of the Tri-Herbal Formula revealed multiple distinct peaks corresponding to phytochemicals derived from Nigella sativa, Boswellia sacra, and Carica papaya. The HPLC chromatogram revealed 17 distinct phytochemical peaks with strong absorbance at 254 nm (Fig. 2) Three diagnostic marker compounds thymoquinone (RT 4.7 min), quercetin (RT 9.2 min), and 11-keto-β-boswellic acid (RT 12.8 min) were quantified at 15.6±1.2, 13.8±1.1, and 18.2±1.4 mg/g dry extract, respectively, yielding a cumulative marker content of 47.6±3.7 mg/g. Batch-to-batch reproducibility demonstrated minimal variation (RSD<3%), indicating robust extraction and compositional stability. Beyond these markers, HPLC analysis identified 17 major phytochemicals, including monoterpenes, phenolic acids, flavonoids, alkaloids, triterpenoids, and isothiocyanates (Table 3). Their retention times ranged from 3.9 to 20.3 min, with quantitative yields between 2.8 and 19.5 mg/g dry extract. Together, these constituents reflect the chemical complexity of THF and highlight its broad phytopharmacological potential.
Comet assay: DNA damage and repair profile: The DNA integrity was quantified using the alkaline comet assay. Representative micrographs are shown in Fig. 3a-e. NMU induction caused extensive DNA strand breaks, evident as elongated comet tails with increased tail DNA % and tail moment in Group B relative to control. Cisplatin produced only partial attenuation, consistent with its own genotoxic potential as a DNA-crosslinking agent.
Quantitative analysis (Table 3) revealed significant inter-group variation for tail DNA %, tail moment, and olive moment (F = 5.76-4.01; p<0.05). The tri-herbal formulation decreased all indices of DNA damage in a concentration-dependent fashion, with the 200 mg/kg and 400 mg/kg groups showing 25-35 % lower tail DNA % compared with NMU+cisplatin (p<0.05). This reduction in comet parameters reflects enhanced DNA repair and chromatin stability (Table 4).

|
| Table 3: | Quantitative phytochemical composition of the Tri-Herbal Formula (THF) | |||
| No. | Compound | Retention time (min) | Quantity (mg/g dry extract) |
| 1 | α-Pinene | 3.9 | 2.8±0.3 |
| 2 | Thymoquinone | 4.7 | 15.6±1.2 |
| 3 | Thymohydroquinone | 5.2 | 9.8±0.7 |
| 4 | p-Cymene | 6.1 | 6.3±0.5 |
| 5 | Carvacrol | 7 | 7.4±0.6 |
| 6 | Caffeic acid | 7.1 | 12.7±1.0 |
| 7 | Chlorogenic acid | 8.6 | 11.3±0.9 |
| 8 | Nigellidine | 8.8 | 4.1±0.4 |
| 9 | Quercetin | 9.2 | 13.8±1.1 |
| 10 | Kaempferol | 10.5 | 11.5±0.8 |
| 11 | Rutin | 11.9 | 10.6±0.9 |
| 12 | 11-Keto-β-Boswellic acid (KBA) | 12.8 | 18.2±1.4 |
| 13 | Acetyl-11-Keto-β-Boswellic acid (AKBA) | 13 | 19.5±1.6 |
| 14 | β-Boswellic acid | 14.2 | 16.7±1.3 |
| 15 | Incensole acetate | 16 | 9.0±0.7 |
| 16 | Lupeol | 18.5 | 8.4±0.6 |
| 17 | Benzyl isothiocyanate | 20.3 | 6.7±0.5 |
| 18 | Papain* | — | — |
| *Papain is a protein enzyme and was excluded from in-silico docking analysis | |||
| Table 4: | Comet assay parameters in mammary tissue (mean±SD, n = 10) | |||
| Parameter | Control (A) | NMU+Cisplatin (B) | Tri-herbal 100 mg/kg (C) |
Tri-herbal 200 mg/kg (D) |
Tri-herbal 400 mg/kg (E) |
F-value | p-value |
| Tail DNA % | 45.13±48.22 | 48.32±48.10 | 32.73±41.64 | 41.98±47.07 | 40.95±46.05 | 5.76 | 0.004* |
| Tail Moment | 12.51±13.72 | 13.60±13.94 | 8.13±11.82 | 10.66±12.85 | 11.44±13.83 | 4.59 | 0.011* |
| Olive Moment | 6.81±8.28 | 7.65±8.21 | 4.85±6.87 | 6.09±7.60 | 6.32±7.75 | 4.01 | 0.019* |
| Statistically significant at p<0.05 (ANOVA+Tukey) | |||||||
Mechanistically, the observed genoprotective effect is attributable to bioactive compounds within the Tri-herbal blend. Nigella sativa seeds are rich in thymoquinone, which quenches reactive oxygen species (ROS) and enhances the expression of base-excision repair enzymes. Boswllia sacra resin contains boswellic acids that inhibit 5-lipoxygenase, reducing lipid peroxidation and inflammatory ROS generation.
|
C. papaya leaf extract provides polyphenols and the cysteine protease papain, which support antioxidant defence and mitochondrial integrity. The combined effect likely promotes DNA repair via Nrf2 activation and p53-dependent cell-cycle arrest, facilitating clearance of damaged cells.
|
| Table 5: | Serum biochemical parameters after six-month treatment with Tri-herbal formula | |||
| Group | Treatment description | ALT (U/L) | AST (U/L) | Urea (mmol/L) | Creatinine (μmol/L) |
| A | Control (untreated) | 32.4±4.1a | 75.8±6.7a | 5.2±0.6a | 58.6±6.2a |
| B | NMU+Cisplatin | 86.9±8.2c | 145.3±10.8c | 10.7±1.3c | 126.4±8.9c |
| C | NMU+Tri-herbal 100 mg/kg | 65.5±6.7b | 118.6±9.3b | 8.5±0.9b | 102.8±7.4b |
| D | NMU+Tri-herbal 200 mg/kg | 49.2±5.3ab | 94.4±8.0ab | 6.9±0.8ab | 84.7±6.8ab |
| E | NMU+Tri-herbal 400 mg/kg | 38.1±4.5a | 81.6±7.1a | 5.8±0.6a | 67.5±5.9a |
| Values are Mean±SD (n = 10), Different superscripts (a-c) within a column indicate statistically significant differences (p<0.05, one-way ANOVA followed by Tukey’s test) | |||||
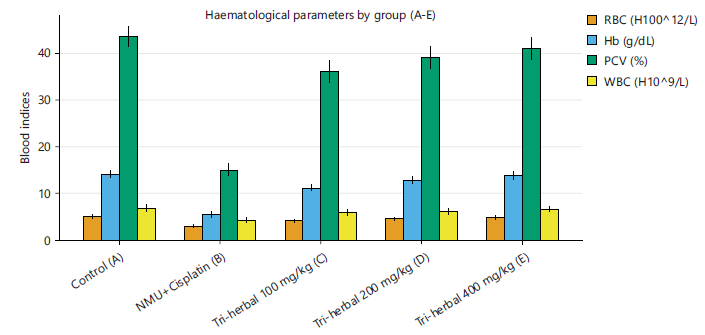
| Table 6: | Haematological indices after six-month treatment with tri-herbal formula | |||
| Group | Treatment description | RBC (×1012/L) | Hb (g/dL) | PCV (%) | WBC (×109/L) | Platelets (×10 /L) |
| A | Control (untreated) | 5.6±0.4a | 14.2±0.8a | 44.0±2.1a | 7.6±0.5a | 392±35a |
| B | NMU+Cisplatin | 2.9±0.3c | 6.0±0.5c | 18.0±1.8c | 4.1±0.4c | 276±28c |
| C | NMU+Tri-herbal 100 mg/kg | 3.8±0.4b | 8.5±0.7b | 26.0±2.0b | 5.0±0.4b | 310±30b |
| D | NMU+Tri-herbal 200 mg/kg | 4.7±0.3ab | 11.8±0.8ab | 36.0±2.2ab | 6.3±0.5ab | 351±33ab |
| E | NMU+Tri-herbal 400 mg/kg | 5.2±0.4a | 13.2±0.7a | 41.0±2.3a | 7.1±0.6a | 384±37a |
| Values are Mean±SD (n = 10), Different superscripts (a-c) within a column indicate statistically significant differences (p<0.05, one-way ANOVA followed by Tukey’s post hoc test) | ||||||
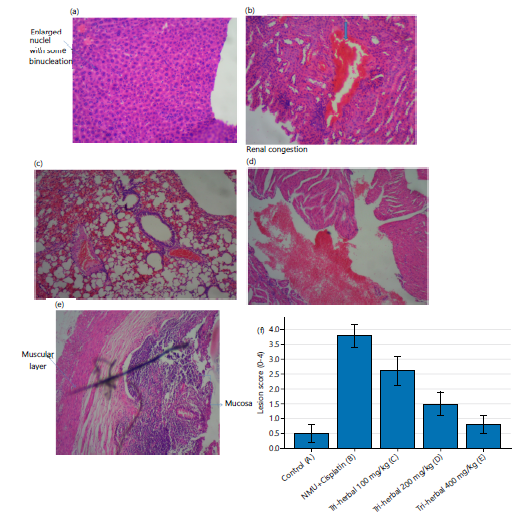
Histopathological alterations: Histopathological assessment of mammary, liver, kidney, lung, and heart tissues provided corroborative evidence for the genotoxic findings (Fig. 4a-f). Control (A): Normal parenchymal and epithelial architecture without congestion or necrosis. NMU+Cisplatin (B): Severe dysplasia in mammary tissue, pulmonary interstitial infiltrates, renal congestion, and dense lymphocytic aggregates. Red blood cells (RBCs) accumulated within vascular sinusoids, indicating haemorrhage and vascular leakage. Tri-herbal 100 mg/kg (C): Moderate inflammatory infiltrates and partial preservation of tissue structure, suggesting limited protection. Tri-herbal 200 mg/kg (D): Reduced necrosis and restoration of epithelial morphology, with mild residual congestion. Tri-herbal 400 mg/kg (E): Nearly normal histoarchitecture, minimal inflammation, and re-established ductal integrity. Semi-quantitative lesion scoring (0 = normal; 4 = severe) (F) showed significant improvement with increasing Tri-herbal dose (F = 8.92, p<0.001). The findings demonstrate a direct correspondence between DNA integrity and tissue regeneration, confirming the restorative influence of the herbal mixture.
Biochemical and haematological evaluation: NMU-induced mice showed significant elevation in ALT, AST, urea, and creatinine compared to control, indicating hepatic and renal dysfunction (Table 5). Treatment with the Tri-herbal formula markedly reduced these enzyme and metabolite levels toward
normal, comparable to the standard drug group suggesting hepatoprotective and nephroprotective effects. However, NMU caused a significant drop in RBC, Hb, and PCV, indicating anaemia (Table 6). These alterations indicate treatment-related bone marrow suppression. Mice receiving the tri-herbal formula displayed a progressive restoration of these indices. The mid and high doses produced values that approached those of the control group (Fig. 5). The tri-herbal formula restored haematological balance, suggesting bone marrow recovery and improved erythropoiesis.
DISCUSSION
Over the six-month experimental period, body-weight monitoring revealed clear dose-dependent effects of the tri-herbal formulation on NMU-induced cachexia. Consistent with prior studies on chemotherapeutic toxicity in rodent breast cancer models9, NMU+cisplatin-treated mice exhibited significant weight loss, poor coat condition, and reduced locomotion. These phenotypes reflect systemic toxicity and metabolic derangements driven by both carcinogen-induced oxidative stress and cisplatin-induced cytotoxicity.
Administration of the tri-herbal formula (Nigella sativa, Boswellia sacra, and Carica papaya leaf) mitigated weight loss in a dose-dependent fashion. High-dose treatment (400 mg/kg/day) restored body weight to near-control levels by week 24, paralleling findings from other polyherbal antioxidant interventions, which suggest enhanced systemic resilience via combined anti-inflammatory and metabolic stabilization effects12. The partial recovery in the 100-200 mg/kg groups indicates that even moderate doses of phytochemicals can ameliorate chemotherapy-induced cachexia, likely through ROS scavenging and mitochondrial protection.
The phytochemical profiling demonstrates a reproducible and chemically diverse formulation, combining terpenoids, flavonoids, and phenolic acids with known anticancer potential. Key markers contribute significantly to its pharmacological activity. The presence of secondary compounds adds antioxidant, anti-inflammatory, and pro-apoptotic support, suggesting synergistic effects. Standardization with low batch-to-batch variability strengthens its translational potential. however, this represents a multi-component therapeutic candidate with pleiotropic mechanisms relevant to oncology.
Mechanistically, the restoration of normal weight likely reflects the synergistic antioxidant and anti-inflammatory activities of the tri-herbal blend. Thymoquinone from N. sativa enhances Nrf2-dependent antioxidant enzyme expression and reduces ROS-mediated muscle protein degradation. Boswellic acids from B. sacra suppress 5-lipoxygenase-mediated lipid peroxidation, while papain and polyphenols in C. papaya leaves improve mitochondrial function and nutrient utilization. Collectively, these bioactivities mirror previously reported effects of combinatorial plant extracts on systemic metabolism in murine cancer models20.
The alkaline comet assay revealed significant genotoxic stress following NMU induction, with cisplatin-treated mice displaying pronounced tail DNA %, tail moment, and Olive Moment values, confirming extensive DNA strand breaks. Similar observations have been reported in N-nitrosomethylurea induced mammary tumour models15, where genotoxic markers correlated with chemotherapeutic-induced oxidative DNA damage.
Tri-herbal treatment significantly reduced all comet parameters in a concentration-dependent manner. High-dose administration (400 mg/kg) reduced tail DNA % by ~25-35% relative to NMU+cisplatin, suggesting enhanced chromatin stability and activation of DNA repair pathways. Mechanistically, this protective effect can be attributed to Thymoquinone from N. sativa neutralizes free radicals and modulates base-excision repair. Boswellic acids inhibit NF-κB and 5-lipoxygenase, reducing oxidative lipid
damage. Cellular repair facilitation of Papaya polyphenols and papain support mitochondrial integrity and p53-mediated DNA damage response, promoting cell-cycle arrest and repair10. These results align with recent reports showing that combinatorial plant extracts can reduce genotoxic burden and improve DNA repair efficiency in chemically induced breast cancer models17.
Histological evaluation corroborated the molecular and systemic observations. NMU+cisplatin induced severe mammary dysplasia, vascular congestion, pulmonary infiltrates, and renal lymphocytic aggregates. In contrast, tri-herbal administration preserved tissue architecture in a dose-dependent manner: Mild residual congestion was observed at 200 mg/kg, while 400 mg/kg restored near-normal morphology. The histopathology supports the comet assay findings, suggesting that improved DNA integrity translates into structural and functional tissue preservation. These observations echo reports in high-impact literature demonstrating that antioxidant-rich botanical formulations reduce chemotherapeutic tissue toxicity while promoting regenerative responses in multiple organs9,14.
Biochemical and haematological evaluations provide critical insights into the systemic and hepatic responses to NMU-induced breast carcinogenesis and the subsequent modulatory effects of the tri-herbal formulation. A marked elevation in serum ALT and AST activities was observed in the NMU+Cisplatin (Group B) compared with the Control (Group A), indicating hepatocellular injury caused by NMU-induced oxidative stress and the cytotoxic effect of cisplatin. This is consistent with previous studies reporting transaminase elevations following exposure to NMU and chemotherapeutic agents due to lipid peroxidation and mitochondrial dysfunction17. Treatment with the tri-herbal formulation at graded doses (100-400 mg/kg) produced a dose-dependent reduction in ALT and AST levels, with Group E (400 mg/kg) showing values approaching normal control. This suggests that the phytoconstituents within the extract, possibly thymoquinone, boswellic acids, and papaya-derived antioxidants, conferred hepatoprotective effects by stabilizing cell membranes and enhancing antioxidant enzyme activities11,13. Similarly, serum urea and creatinine levels, biomarkers of renal function, were significantly elevated in Group B, reflecting nephrotoxicity associated with NMU and cisplatin. Administration of the tri-herbal extract mitigated this effect, with the 200 and 400 mg/kg doses restoring renal indices toward normal. These findings align with those of Saetang et al.16, who reported that Carica papaya extract ameliorates renal oxidative stress in tumour-bearing models.
Haematological assessment revealed significant reductions in RBC count, haemoglobin concentration, and platelets, coupled with elevated WBC count in Group B compared with the control. These alterations signify anaemia and systemic inflammatory response, which are typical in NMU-induced carcinogenesis22. The administration of the tri-herbal formulation significantly reversed these haematological perturbations in a dose-dependent manner, with Group E showing near-normal values. This improvement may be attributed to the bioactive phytochemicals promoting erythropoiesis, antioxidant protection of erythrocyte membranes, and normalization of bone marrow activity12,15.
Moreover, the tri-herbal formula exerts multifactorial protective effects such as attenuation of cachexia and weight loss, reduction of DNA strand breaks, enhanced repair, and preservation of histoarchitecture, reduced inflammation and haemorrhage. Mechanistically, these effects appear to converge on the modulation of oxidative stress, DNA repair activation (Nrf2, p53), and inflammatory signalling. The dose-dependent efficacy highlights the translational potential of standardized herbal formulations as adjuvants in breast cancer therapy, mitigating chemotherapeutic toxicity while enhancing DNA repair capacity.
Finally, this study provides compelling evidence that the Tri-herbal blend of N. sativa, B. sacra, and C. papaya leaves exerts systemic, genoprotective, and histo-restorative effects in NMU-induced murine breast cancer. These findings complement prior high-impact research and support further mechanistic and clinical investigations into polyherbal adjuvants for cancer management.
CONCLUSION
The study shows that a tri-herbal formulation of Nigella sativa, Boswellia sacra, and Carica papaya effectively protects against NMU-induced mammary cancer in mice. Treatment improved body weight, restored tissue structure, and reduced DNA damage in a dose- dependent manner. Histopathological and biochemical evaluations confirmed the protection of vital organs and enhancement of DNA integrity. The interactive bioactive ingredients are likely to act through the antioxidant, anti-inflammatory, and DNA repair activities involving Nrf2, p53, and NF-κB modulation. However, the tri-herbal preparation was found to have significant drug efficacy and safety, suggesting its promise as an auxiliary approach for the therapy of breast cancer, subject to further molecular and drug action characterization and verification.
SIGNIFICANCE STATEMENT
This study demonstrates that a tri-herbal formulation of Nigella sativa, Boswellia sacra, and Carica papaya significantly attenuates tumour progression and genotoxic damage in NMU-induced mammary carcinogenesis. The findings highlight its potential as a complementary phytotherapeutic strategy with antioxidative and DNA-protective effects, offering a safer alternative or adjunct to conventional chemotherapy in breast cancer management.
REFERENCES
- Letafati, A., Z. Taghiabadi, N. Zafarian, R. Tajdini and M. Mondeali et al., 2024. Emerging paradigms: Unmasking the role of oxidative stress in HPV-induced carcinogenesis. Infect. Agents Cancer, 19.
- Triantaphyllopoulos, K.A., N.D. Ragia, M.C.E. Panagiotopoulou and T.G. Sourlingas, 2025. Integrating inflammatory and epigenetic signatures in IBD-associated colorectal carcinogenesis: Models, mechanisms, and clinical implications. Int. J. Mol. Sci., 26.
- Su, L.J., J.H. Zhang, H. Gomez, R. Murugan and X. Hong et al., 2019. Reactive oxygen species-induced lipid peroxidation in apoptosis, autophagy, and ferroptosis. Oxid. Med. Cell. Longevity, 2019.
- Dastjerdi, M.N., E.M. Mehdiabady, F.G. Iranpour and H. Bahramian, 2016. Effect of thymoquinone on P53 gene expression and consequence apoptosis in breast cancer cell line. Int. J. Preventive Med., 7.
- Liang, X., J. Weng, Z. You, Y. Wang and J. Wen et al., 2025. Oxidative stress in cancer: From tumor and microenvironment remodeling to therapeutic frontiers. Mol. Cancer, 24.
- Ngo, V. and M.L. Duennwald, 2022. Nrf2 and oxidative stress: A general overview of mechanisms and implications in human disease. Antioxidants, 11.
- Fakheri, H., M.A. Sajadi, A. Afrashteh, W. Ma, L. Xia and M. Valilo, 2023. The nuclear factor erythroid 2-related factor 2/p53 axis in breast cancer. Biochem. Med., 33: 266-278.
- Sajadimajd, S. and M. Khazaei, 2018. Oxidative stress and cancer: The role of Nrf2. Curr. Cancer Drug Targets, 18: 538-557.
- Reuter, S., S.C. Gupta, M.M. Chaturvedi and B.B. Aggarwal, 2010. Oxidative stress, inflammation and cancer: How are they linked? Free Radical Biol. Med., 49: 1603-1616.
- Jehan, S., C. Zhong, G. Li, S.Z. Bakhtiar, D. Li and G. Sui, 2020. Thymoquinone selectively induces hepatocellular carcinoma cell apoptosis in synergism with clinical therapeutics and dependence of p53 status. Front. Pharmacol., 11.
- Woo, C.C., A.P. Kumar, G. Sethi and K.H.B. Tan, 2012. Thymoquinone: Potential cure for inflammatory disorders and cancer. Biochem. Pharmacol., 83: 443-451.
- Esmaeili, F., T. Lohrasebi, M. Mohammadi-Dehcheshmeh and E. Ebrahimie, 2021. Evaluation of the effectiveness of herbal components based on their regulatory signature on carcinogenic cancer cells. Cells, 10.
- Siddiqui, M.Z., 2011. Boswellia serrata, a potential antiinflammatory agent: An overview. Indian J. Pharm. Sci., 73: 255-261.
- Al-Bahlani, S., I.A. Burney, B. Al-Dhahli, S. Al-Kharusi, F. Al-Kharousi, A. Al-Kalbani and I. Ahmed, 2020. Boswellic acid sensitizes gastric cancer cells to Cisplatin-induced apoptosis via p53-mediated pathway. BMC Pharmacol. Toxicol., 21.
- Jenča, A., D. Mills, H. Ghasemi, E. Saberian and A. Jenča et al., 2024. Herbal therapies for cancer treatment: A review of phytotherapeutic efficacy. Biol.: Targets Ther., 18: 229-255.
- Saetang, J., A. Tedasen, S. Sangkhathat, N. Sangkaew and S. Dokduang et al., 2022. The attenuation effect of low piperine Piper nigrum extract on doxorubicin-induced toxicity of blood chemical and immunological properties in mammary tumour rats. Pharm. Biol., 60: 96-107.
- Manful, C.F., E. Fordjour, E. Ikumoinein, L. Abbey and R. Thomas, 2025. Therapeutic strategies targeting oxidative stress and inflammation: A narrative review. BioChem, 5.
- Fakhri, K.U., D. Sharma, H. Fatma, D. Yasin and M. Alam et al., 2025. The dual role of dietary phytochemicals in oxidative stress: Implications for oncogenesis, cancer chemoprevention, and ncRNA regulation. Antioxidants, 14.
- Rizeq, B., I. Gupta, J. Ilesanmi, M. AlSafran, M. Mizanur Rahman and A. Ouhtit, 2020. The power of phytochemicals combination in cancer chemoprevention. J. Cancer, 11: 4521-4533.
- Dubey, A. and B. Sharma, 2025. Targeting oxidative stress biomarkers in breast cancer development and the potential protective effect of phytochemicals. Drugs Drug Candidates, 4.
- Baris, M.M., E. Serinan, M. Calisir, K. Simsek and S. Aktas et al., 2020. Xenograft tumor volume measurement in nude mice: Estimation of 3D ultrasound volume measurements based on manual caliper measurements. J. Basic Clin. Health Sci., 4: 90-95.
- Sowunmi, K., O. Nwoye, O. Esther, K. Ayomikun and A. Lawal, 2025. Chemo-preventive potential of ethanolic extract of Nigella sativum on N-nitroso methyl urea (NMU) induced breast cancer in female Albino mice. Cell Biol., 13: 23-29.
- Singh, N.P., M.T. McCoy, R.R. Tice and E.L. Schneider, 1988. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res., 175: 184-191.
- Avram, C. and M. Mărușteri, 2022. Normality assessment, few paradigms and use cases. Rev. Rom. Med. Lab., 30: 251-260
How to Cite this paper?
APA-7 Style
Sowunmi,
K., Ibrahim,
M.O., Ogundele,
O.E. (2026). Chemotherapeutic Potential of a Tri-Herbal Formulation Against NMU-Induced Mammary Tumors via Histological Remodeling and Genoprotection. Trends in Pharmacology and Toxicology, 2(1), 47-59. https://doi.org/10.21124/tpt.2026.47.59
ACS Style
Sowunmi,
K.; Ibrahim,
M.O.; Ogundele,
O.E. Chemotherapeutic Potential of a Tri-Herbal Formulation Against NMU-Induced Mammary Tumors via Histological Remodeling and Genoprotection. Trends Pharm. Toxicol. 2026, 2, 47-59. https://doi.org/10.21124/tpt.2026.47.59
AMA Style
Sowunmi
K, Ibrahim
MO, Ogundele
OE. Chemotherapeutic Potential of a Tri-Herbal Formulation Against NMU-Induced Mammary Tumors via Histological Remodeling and Genoprotection. Trends in Pharmacology and Toxicology. 2026; 2(1): 47-59. https://doi.org/10.21124/tpt.2026.47.59
Chicago/Turabian Style
Sowunmi, Kehinde, Micheal Omeyiza Ibrahim, and Olusegun Emmanuel Ogundele.
2026. "Chemotherapeutic Potential of a Tri-Herbal Formulation Against NMU-Induced Mammary Tumors via Histological Remodeling and Genoprotection" Trends in Pharmacology and Toxicology 2, no. 1: 47-59. https://doi.org/10.21124/tpt.2026.47.59

This work is licensed under a Creative Commons Attribution 4.0 International License.




